About us
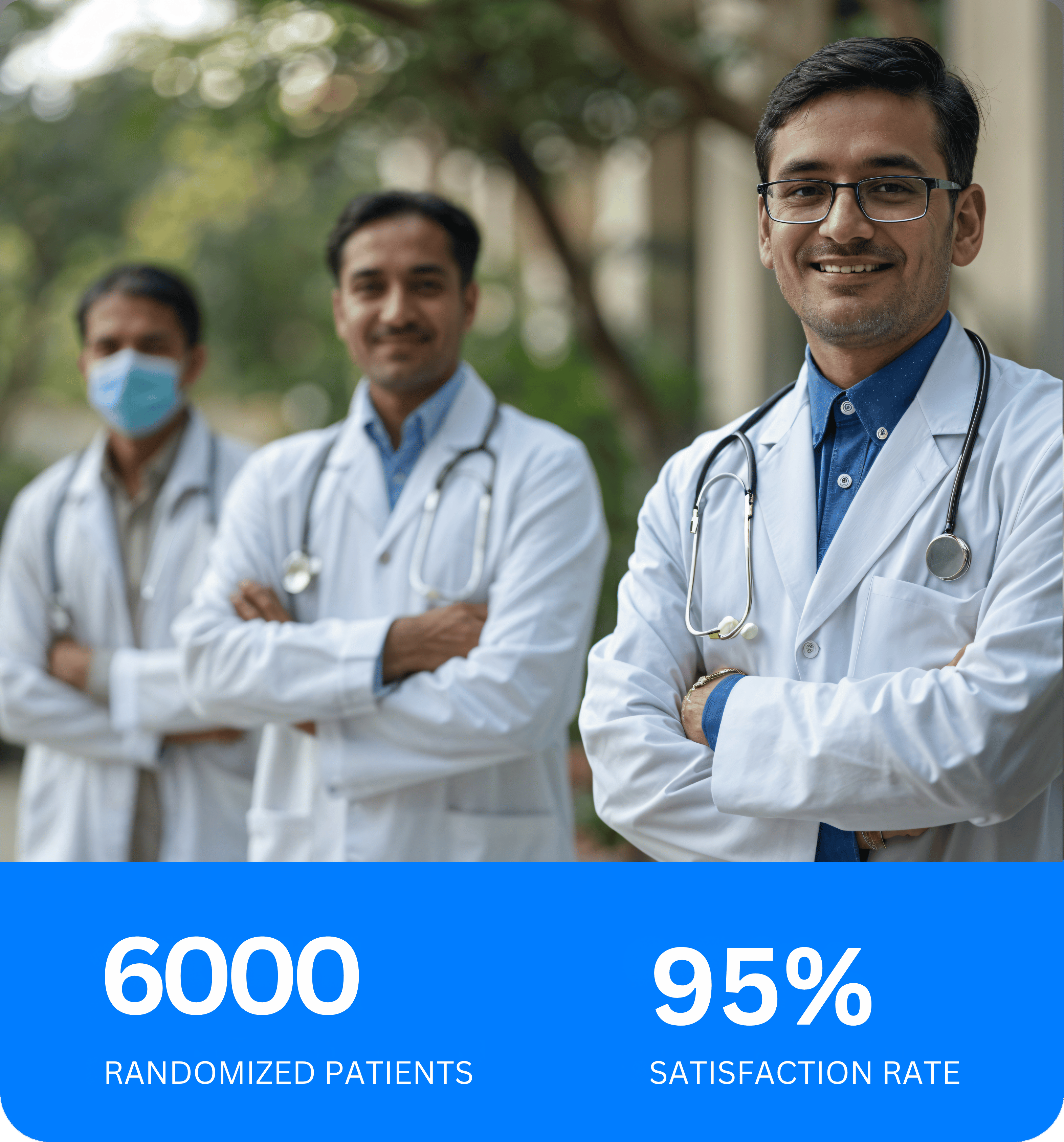
Since 2020 we are providing best clinical trial services to sponsor, CRO and principal investigators.
We’re Putting your Comfort List
Site selection and activation
Study start-up support
Patient recruitment and retention
Clinical monitoring
Timely query resolution
Data management
Frequent training to avoid PD/PV
Internal auditing prior to monitoring
About us
Oxygen clinical research and services is a site management organization (smo) offering comprehensive site management services to contract research organizations (cro) and pharmaceutical & / or biotechnology companies.
Sole purpose of ACR is to support the successful outcome of all clinical trials with time line and cost effectiveness. We are here to provide you with ethical, quality oriented, enthusiastic, and experienced clinical research sites in an expedited and efficient manner.
Our vision
-
Oxygen clinical research and services is a leading site management organization (SMO) dedicated to providing comprehensive site management services to contract research organizations (CROs) and pharmaceutical companies.
-
Our primary objective is to provide timely and cost-effective support for clinical trials.
-
As clinical research sites, we pride ourselves on providing honest, quality-oriented, enthusiastic, and experienced services in a timely an efficient manner. an expedited and efficient manner.
Our mission
-
The quality of our work will never be compromised, and our projects will be delivered according to the deadlines.
-
As a good clinical practice-compliant and subject-centred organization, we are committed to contributing to a healthy society.
-
To create a healthy work environment for our employees while striving for excellence.
What we do ?
Oxygen Clinical Research is a network of high-performing principal investigators. We realize that successful outcome of clinical trials above all depends on the skills and dedication of the chosen physicians we work with. Our investigators have successfully completed 25+ clinical trials. We conduct a comprehensive range of clinical trials, including Phase I to IV studies, bioavailability and bioequivalence assessments, medical device trials, and post-marketing observational studies, all aimed at advancing research across diverse therapeutic areas..
Oxygen Clinical Research supports its Principal Investigators by:
- Promotion of Investigator and Investigative site: Oxygen Clinical Research manages and executes a full therapeutic portfolio on your behalf. Oxygen Clinical Research represents you and your clinic before sponsor during the site selection process. Our specialists ensure that your investigative site is “armed” with the proper knowledge and equipment to succeed in a highly competitive pharmaceutical environment.
- Screening Study Opportunities, Site Feasibility Completion & Submission: Before approaching you with a new study Oxygen Clinical Research performs initial assessment of your center’s individual needs, access to the adequate pool of patients, and the infrastructure for conducting the clinical trials.
- Training, Training Materials: Oxygen Clinical Research takes responsibility for ensuring research staff is trained in the latest regulatory requirements and the most effective tools to implement GCP at your research center.
- Placement of Qualified and trained research staff: Oxygen Clinical Research in consists of qualitatively highly trained professionals for managing clinical trials: Study Coordinators and data entry personnel.
- Standardization of research processes: The innovative and standardized approach will allow your center to handle multiple studies simultaneously.
- Provision of Investigator Site SOPs: Enhance the quality of clinical research in your research center by standardizing your procedures with the help of SOPs for Sites. With SOPs for sites the quality of the research will be warranted throughout all levels within your research center, because all involved parts will start to work in a uniform matter. Help your research center to meet the high-quality demands sponsors request.
- Regulatory Documents Preparation/Submissions: Oxygen Clinical Research takes care of the preparation and submission of all Regulatory Documentation to the Independent Ethics Committee, CRO, Sponsor and other strategic partners. Adequate research amenities on site: Oxygen Clinical Research provides the research center with all required equipment directed by study protocol (e.g. centrifuge, spirometry, ECG, Heigh scale, weight machine, BP Apparatus, thermohydrometer, etc.).
- Contract Review, Negotiation Process– The Study Agreement (CTA) is often skimmed-over by investigators, who are often too busy to afford the time to read pages of legalese. Since you are part of Oxygen Clinical Research network we consider ourselves your legal representative in these agreements.
- Source Document Worksheet Creation: Oxygen Clinical Research develops study related documents. These worksheets facilitate information management at our sites and help to ensure the accuracy and completeness of data in the CRFs.
- Recruitment Support: Oxygen Clinical Research in collaboration with Investigators are able to recruit large numbers of subjects in a short period of time. Approaching and identifying trial subjects is often time consuming. Oxygen Clinical Research has developed a specific subject selection procedure for clinical trials. Potential patients are informed about the trial through an information campaign: advertisement, referral program etc.
- Payment Administration: Tracking & Reconciliation, Timeliness, Advocacy: Oxygen Clinical Research negotiates budget discussing with principal integrator. Before launching new trial, Oxygen Clinical Research estimates study costs and performs complete marketing analysis. Oxygen Clinical Research tracks the enrolment progress for you and calculates the monies due, based on the procedures/visits performed, and reconciles this data with payments received from sponsors to ensure accuracy and timeliness of all study payments. Oxygen Clinical Research closely monitors payments on your behalf and ensures that payments available for you according to the schedule and there are no payment delays. Feasibility And Site Selection Activities: Before approaching Sponsor/ CRO to Research Center Oxygen Clinical Research performs initial assessment of the center’s individual needs, access to the adequate pool of patients, and the infrastructure for conducting the clinical study. By thorough analysis Oxygen Clinical Research evaluates the center’s potential to succeed in the study. This approach will save your time and allow us to prevent number of issues in the later stages of the study and results in 100% success in the performance of your clinic.
- Training and Professional Development: Oxygen Clinical Research takes responsibility for ensuring research staff is trained in the latest regulatory requirements and have the most effective tools to implement ICH/GCP guidelines. Also, Oxygen Clinical Research ensures that site personnel are fully trained on protocol and study related material. Oxygen Clinical Research training programs are available for Clinical Research Coordinators and Investigators. These 1-2 day programs describe all aspects of the clinical trial and review the roles and responsibilities of all members of the research team. Contact us online for course availability and pricing information.
- “Task oriented” Approach: The “task oriented” approach will bring to your study consistency and efficiency. Each team member at Oxygen Clinical Research is responsible for the certain task. For example, data entry professionals are specializing to enter data only and do not deal with other aspects of your trial.
- Regulatory Documents Preparation/Submissions: Oxygen Clinical Research takes care of the preparation and submission of all Regulatory Documentation to the Independent Ethics Committee, CRO, Sponsor and other strategic partners.
- Timeline Tracking: Oxygen Clinical Research will identify and assist clients in establishing and maintaining study milestones and deadlines throughout the conduct of the clinical trial.
- Source Document Worksheet Creation: Oxygen Clinical Research develops study related documents. These worksheets facilitate information management at our sites and help to ensure the accuracy and completeness of data in the CRFs.

