Decentralized Clinical Trials (DCTs): What They Mean for Indian Sites in 2025
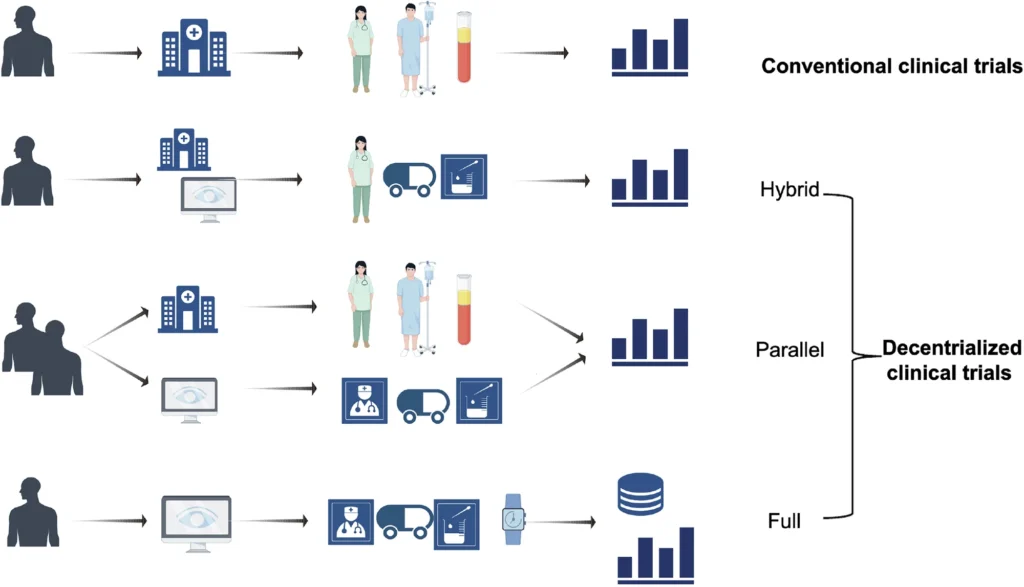
Decentralized Clinical Trials India. In recent years, the world of clinical research has witnessed a rapid transformation of trial design and conduct. To begin with, decentralized clinical trials (DCTs) leverage digital health technologies, remote monitoring, telemedicine, and home-based assessments to bring trial activities directly to participants rather than requiring travel to centralized sites. In other words, this model shifts the focus from site-centric to patient-centric research. As we approach 2025, DCTs are no longer experimental add-ons but are becoming integral to drug development globally. click here Moreover, their adoption is accelerating due to advancements in digital infrastructure and patient expectations. For Indian research sites, this shift presents both exciting opportunities and unique challenges. Therefore, adapting to this evolving model is becoming increasingly important for long-term success. This article explores how DCTs are reshaping the Indian clinical trial landscape, what students and emerging professionals need to know, and how sites can prepare for the coming wave of decentralized research. ConclusionDecentralized Clinical Trials are poised to redefine the conduct of research in India by 2025. For trial sites, this evolution presents a dual mandate: embrace digital transformation to seize new opportunities, and address the unique operational and regulatory challenges of a diverse, rapidly changing landscape. Students and emerging professionals who cultivate specialized skills in DCT design, technology management and cross-cultural engagement will find themselves at the forefront of a dynamic and socially impactful field. As India’s research ecosystem aligns with global best practices, decentralized trials promise greater inclusivity, efficiency and patient-centricity—paving the way for faster delivery of innovative therapies to millions in need. click here