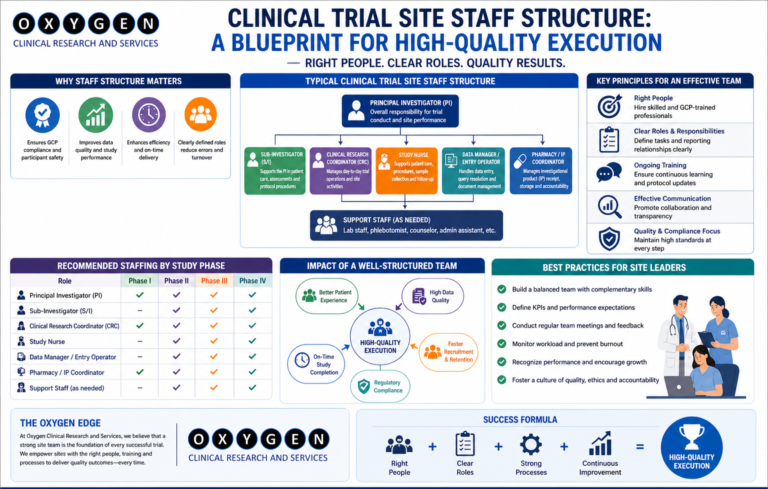
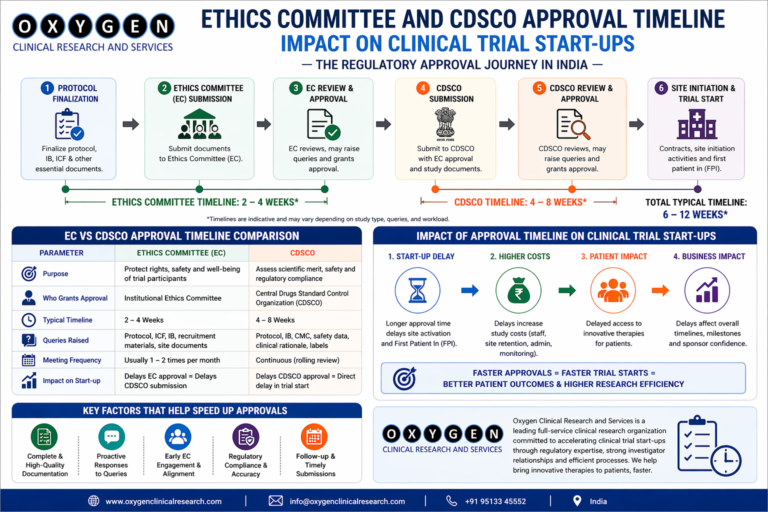
Clinical Trial Site Qualification Visit (SQV) Process: What Sponsors Evaluate
Clinical Trial SQV Process. I have spent fifteen years standing in hospital corridors, sitting across from overworked Principal Investigators (PIs), and reviewing site files that looked perfect on paper but were a liability in reality. I have seen trials delayed by six months because a site was qualified based on “reputation” rather than operational reality. I have seen data get tossed during a CDSCO audit because a site’s source documentation didn’t meet ALCOA+ standards, despite a “successful” SQV. A Site Qualification Visit (SQV) is frequently treated as a box-ticking exercise by junior monitors or rushed CROs. This is a mistake that costs sponsors millions. In India’s unique regulatory and clinical landscape, the SQV is the only barrier between a successful trial and a multi-year regulatory nightmare. If you don’t find the cracks during the SQV, you will find them during the first patient audit. Executive Summary: The Sponsor Perspective on Risk For a Sponsor or a Clinical Operations head, the SQV is about three things: predictability, compliance, and ROI. Every day a site is stagnant costs thousands of dollars. In India, the “New Drugs and Clinical Trials Rules (2019)” have streamlined some aspects, but the complexity of Ethics Committee (EC) management and investigator commitment remains a bottleneck Clinical Trial SQV Process. What is at stake during an SQV? If a site fails at the SQV stage, it is a win. The real failure is qualifying a site that should never have been selected in the first place. The Site Qualification Process: Beyond the Facility Tour When we walk into a site for an SQV in India, we are looking for a culture of compliance. Most PIs will show you their high-end diagnostic equipment or their CV. As a seasoned leader, I look at the study coordinator’s desk and the site’s Standard Operating Procedures (SOPs) Clinical Trial SQV Process. The PI Interview: Assessing Real Commitment Many Indian PIs are “academic giants” but “operational ghosts.” During the SQV, I evaluate if the PI is actually going to oversee the trial or if they are delegating everything to a junior resident. Staffing and Infrastructure We evaluate the Site Management Organization (SMO) support if applicable. If a site relies on a single coordinator for three different therapeutic areas, your data quality will suffer. We check for: Where the Delays Happen: The Indian Context Indian clinical trials often stall at the transition between SQV and Site Initiation Visit (SIV). The SQV must identify these potential “silent delays”: Learn more about navigating clinical research services in India Real Operational Insights: What Actually Works vs. What Fails Sites often fail not because they lack equipment, but because they lack process. In my experience, a Tier-2 city hospital with a dedicated PI and a meticulous coordinator often outperforms a “prestigious” metro hospital where the PI is rarely on-site. Evidence of Recruitment Capability During an SQV, don’t just ask for a recruitment estimate. Ask to see the de-identified patient logs from the last six months. If a site says they see 50 diabetic patients a week but cannot show a log to prove it, they are guessing. Guessing leads to recruitment failure. ALCOA+ Compliance Data must be Attributable, Legible, Contemporaneous, Original, Accurate, and now Complete, Consistent, Enduring, and Available. I have seen sites using scrap paper for primary vitals then “transcribing” them into source notes. This is a red flag. During the SQV, we must verify that the site has a system for capturing data at the point of care. Case Studies: Lessons from the Field Case Study 1: The Celebrity PI Trap Case Study 2: The Cold Chain Crisis Case Study 3: The Unregistered Ethics Committee Case Study 4: The Recruitment Overestimate Challenges and Mitigation: The Unfiltered Reality Executing trials in India involves navigating a mix of high-tech facilities and legacy bureaucratic hurdles. Mitigation: Ensure the site has a documented internal training SOP. Don’t just train the coordinator; train the site. Mitigation: Ask the PI how they handle SAE (Serious Adverse Event) reporting. If their response is “the CRO handles it,” they are a liability. Mitigation: Verify NABL/CAP certifications during the SQV or plan for a central lab. Our site management expertise addresses these specific risks Myths vs. Reality Reality: While the bureaucracy is real, government hospitals in India often have the highest patient volumes and the most loyal patient pools for long-term follow-up studies. Reality: A certificate is a piece of paper. During an SQV, I ask the staff to explain the Informed Consent Form (ICF) process for an illiterate patient. Their answer tells me more than any certificate. Reality: In many Indian hospitals, EMRs are used for billing, not for clinical notes. You must check if the clinical source data is actually in the EMR or still in handwritten folders. Common Mistakes Sponsor Mistakes CRO Mistakes Site Mistakes Counterintuitive Insight: Why “Busy” PIs Can Be a Red Flag Most sponsors want the top-ranked doctor in the country. In India, these doctors often see 100+ patients a day. They have no time to read the protocol, let alone oversee the daily nuances of a complex Phase II study Clinical Trial SQV Process. The best-performing sites are often led by “Mid-Career” PIs. These are investigators who are established enough to have patient volume but are still hungry enough to be personally involved in the research. They value the publication potential and the data quality. During the SQV, I look for the PI who asks me technical questions about the drug’s mechanism of action. That shows real interest Clinical Trial SQV Process. Practical Sponsor Checklist Feasibility Stage (Pre-SQV) Startup Stage (During SQV) Execution Stage (Post-SQV Assessment) Regulatory and Compliance Context Navigating the Indian regulatory environment requires adherence to multiple bodies: For technical inquiries or site-specific feasibility in India, you can reach out directly at govindpawar@oxygenclinicaltrials.com or connect via LinkedIn. Suggested Visuals for Deployment External References