The clinical research landscape is complex, demanding, and highly regulated. For Contract Research Organizations (CROs) navigating the intricacies of clinical trials in a diverse and dynamic region like Maharashtra, India, the selection of the right Site Management Organization (SMO) is paramount. A strong SMO acts as an extension of the CRO, ensuring trials are conducted efficiently, ethically, and in compliance with all applicable regulations.
The Vital Role of SMOs in Clinical Research
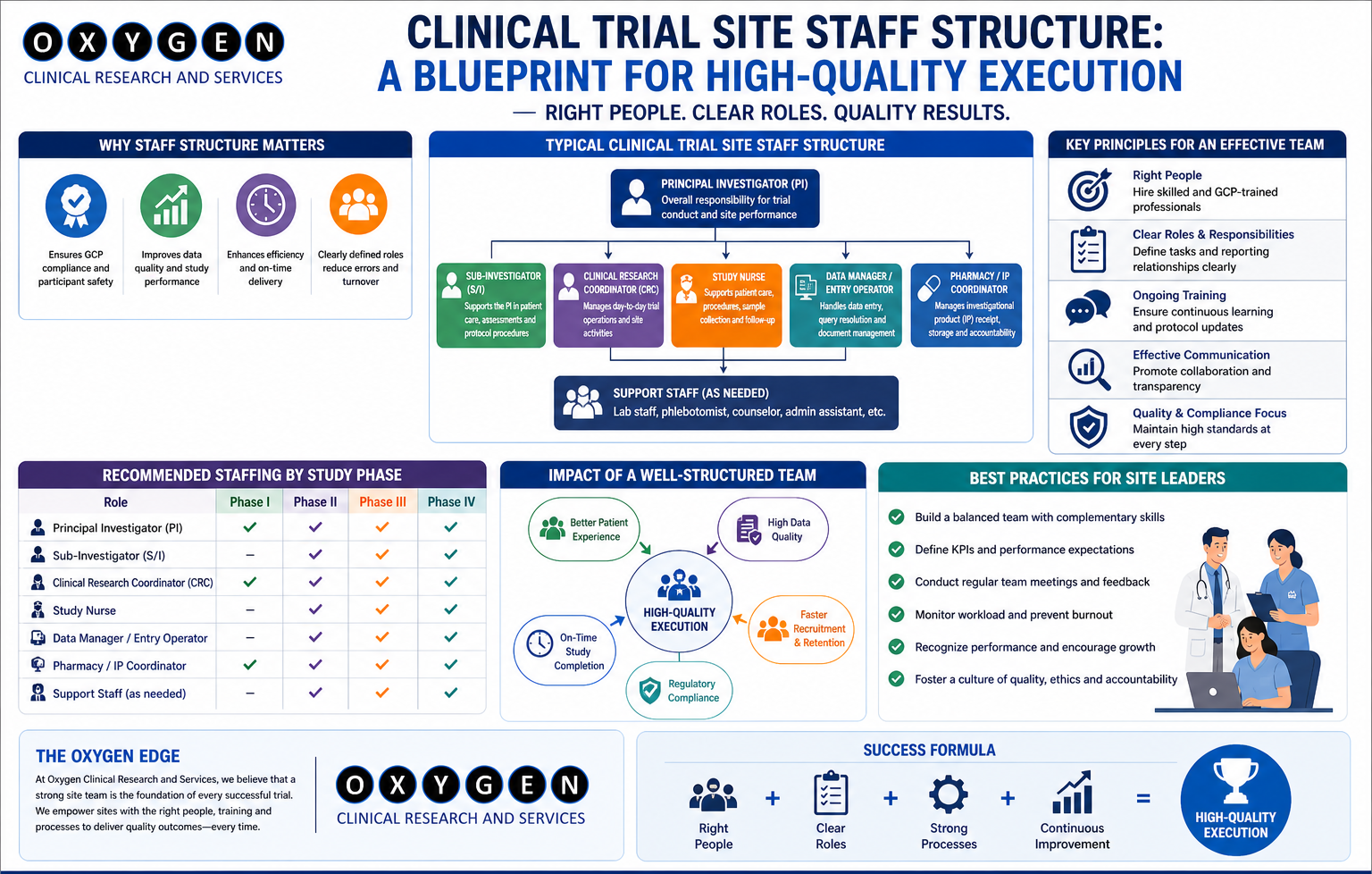
Think of SMOs as the boots on the ground for clinical trials. They bridge the gap between CROs, pharmaceutical companies, and the actual clinical trial sites (hospitals, clinics, and research institutions). SMOs assume a wide range of responsibilities, alleviating the burden on both the CRO and the site staff.
Here’s a glimpse into what a good SMO typically handles:
- Site Identification and Selection: Identifying sites that align with the study protocol and have the necessary resources and patient population.
- Regulatory Compliance: Ensuring all activities adhere to local and international regulations (e.g., GCP guidelines, Indian regulations).
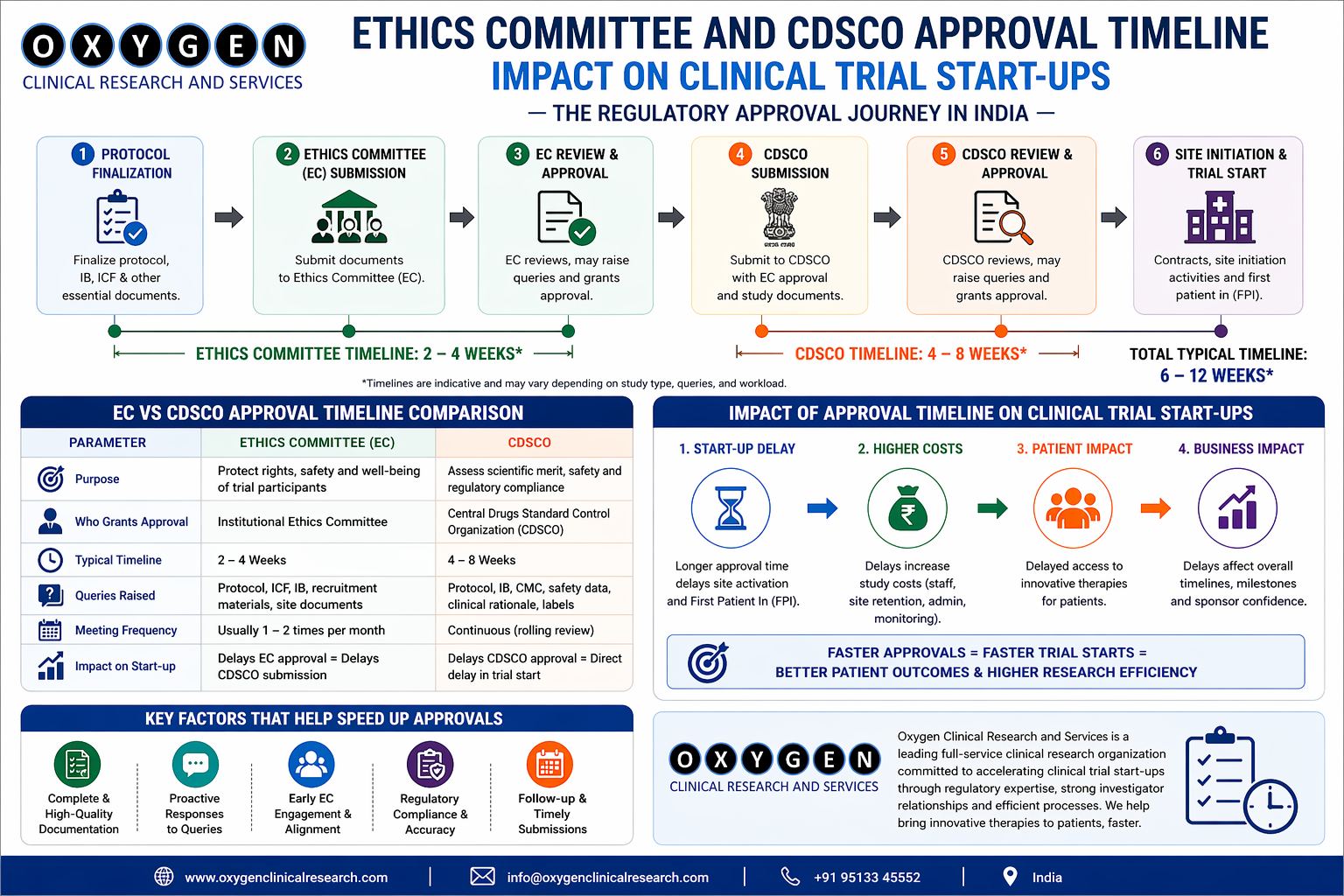
- Ethics Committee Submissions: Preparing and submitting documents to ethics committees for approval.
- Patient Recruitment and Retention: Developing and implementing strategies to attract and retain eligible patients for the trial.
- Data Management: Ensuring accurate and timely data collection, entry, and management.
- Quality Assurance: Monitoring site activities to ensure adherence to the protocol and GCP guidelines.
- Training: Providing training to site staff on the protocol, data collection procedures, and regulatory requirements.
- Budget Management: Overseeing the study budget and ensuring proper financial management at the site level.
- Contract Negotiation: Assist in contract negotiation with sites
Why Maharashtra? A Hub for Clinical Research
Maharashtra has emerged as a significant hub for clinical research in India due to several factors:
- Large and Diverse Patient Population: The state offers access to a diverse patient pool, crucial for clinical trials across various therapeutic areas.
- Advanced Medical Infrastructure: Maharashtra boasts a well-established network of hospitals, research institutions, and medical professionals.
- Government Support: The state government actively promotes clinical research through various initiatives and policies.
- Skilled Workforce: A large pool of trained medical professionals, researchers, and technicians are available.
- Strategic Location: Maharashtra’s connectivity and accessibility make it an attractive location for conducting clinical trials.
Key Considerations When Selecting an SMO in Maharashtra
Choosing the right SMO can significantly impact the success of a clinical trial. CROs should carefully evaluate potential partners based on the following criteria:
- Experience and Expertise: Look for an SMO with a proven track record in the specific therapeutic area and phase of the clinical trial.
- Regulatory Knowledge: Ensure the SMO has a thorough understanding of Indian regulations and GCP guidelines.
- Network of Sites: A strong network of well-established and reputable clinical trial sites is crucial.
- Quality Management System: A robust quality management system ensures adherence to ethical and regulatory standards.
- Communication and Transparency: Excellent communication and transparency are essential for effective collaboration.
- Technology and Data Management: The SMO should have the necessary technology and expertise to manage data efficiently and securely.
- Patient Recruitment Capabilities: A proven track record of successful patient recruitment and retention is a significant advantage.
- Financial Stability: Ensure the SMO is financially stable and capable of supporting the clinical trial throughout its duration.
The Importance of Due Diligence
This list is not exhaustive, and it’s crucial for CROs to conduct thorough due diligence before selecting an SMO. This includes:
- Requesting proposals and conducting interviews: Evaluate the SMO’s capabilities and experience in detail.
- Checking references: Speak with other CROs and sponsors who have worked with the SMO.
- Visiting potential sites: Assess the SMO’s relationship with its partner sites and their capabilities.
- Reviewing the SMO’s quality management system: Ensure it meets industry standards and regulatory requirements.
- Assessing financial stability: Verify the SMO’s financial health and ability to support the trial.
The Future of SMOs in Maharashtra
The clinical research landscape is constantly evolving, and SMOs must adapt to meet the changing needs of CROs and pharmaceutical companies. Key trends shaping the future of SMOs in Maharashtra include:
- Increased focus on technology: SMOs are increasingly adopting technology to improve efficiency, data quality, and patient engagement.
- Emphasis on patient-centricity: SMOs are focusing on improving the patient experience and ensuring patient retention.
- Greater collaboration: SMOs are collaborating more closely with CROs and sponsors to streamline clinical trial processes.
- Expansion into new therapeutic areas: SMOs are expanding their expertise into emerging therapeutic areas such as gene therapy and personalized medicine.
Conclusion
Choosing the right SMO is a critical decision for CROs conducting clinical trials in Maharashtra. By carefully evaluating potential partners based on experience, expertise, regulatory knowledge, and other key criteria, CROs can significantly increase their chances of success. The top SMO companies for CROs in Maharashtra play a vital role in ensuring that clinical trials are conducted efficiently, ethically, and in compliance with all applicable regulations, ultimately contributing to the advancement of medical knowledge and the development of new therapies.
“The key to successful clinical trials lies in strong partnerships and a commitment to quality at every stage of the process.”