Clinical Trial Site Selection. A sponsor recently approached me after a Phase III global study stalled. They had selected ten high-profile oncology sites in India based on impressive feasibility questionnaires. Six months post-SIV, four sites had zero enrollments. The “paper patients” promised during feasibility didn’t exist in the actual outpatient clinics, and the Principal Investigators (PIs) were over-committed to five other competing global trials. This isn’t a unique failure; it is the standard outcome of selecting sites based on marketing rather than operational reality.
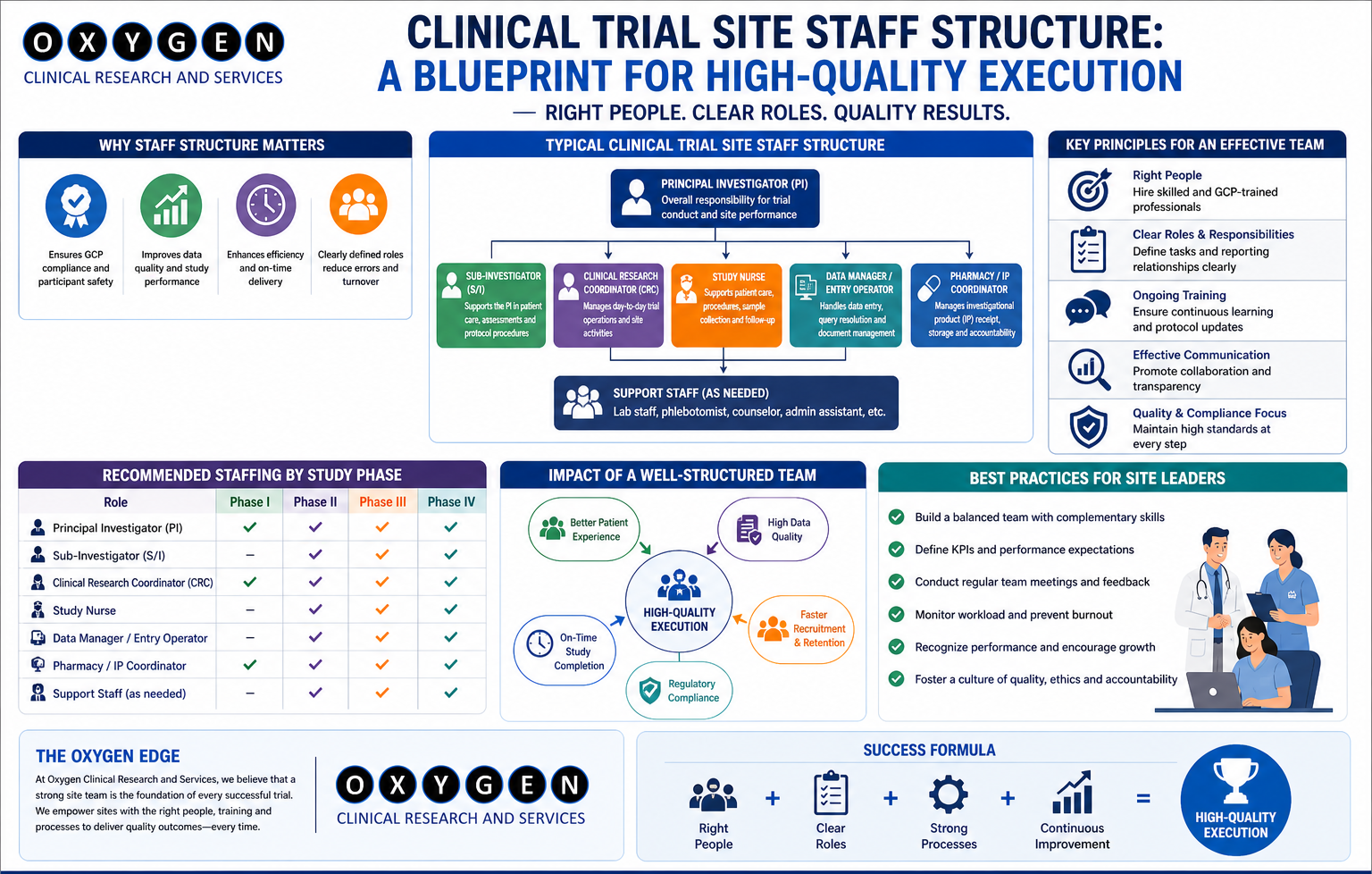
Clinical Trial Site Selection In India, site selection is the most significant risk-mitigation step in the study lifecycle. If you get the site wrong, no amount of monitoring or “rescue” activity will bring the timeline back. You are not just looking for a site with a freezer and a centrifuge; you are looking for an ecosystem that can survive a CDSCO audit, meet recruitment targets without compromising GCP, and manage the administrative burden of the New Drugs and Clinical Trials Rules (2019).
Executive Summary: Operational Site Comparison
Clinical Trial Site Selection Selecting a site based only on the PI’s CV is a recipe for delay. Sponsors must evaluate the infrastructure, the institutional ethics committee (IEC) efficiency, and the actual availability of the study coordinator.
Table 1: Site Selection Framework and Impact Analysis
| Sr. No. | Site Category | Average Startup (Weeks) | Recruitment Reliability | Data Quality Rating | Regulatory Audit Risk | Cost per Patient | Staff Turnover | EC Meeting Frequency | PI Involvement |
| 1 | Large Tier-1 Corporate | 18–24 | High Volume | Moderate | Low | High | High | Monthly | Low |
| 2 | Govt. Academic Center | 26–40 | Very High | Variable | Moderate | Low | Low | Quarterly | Moderate |
| 3 | Private Specialist Clinic | 12–16 | Consistent | High | Low | Moderate | Low | Monthly | High |
| 4 | Multi-Specialty Hospital | 16–20 | Moderate | High | Low | Moderate | Moderate | Bi-Monthly | Moderate |
| 5 | Regional Cancer Center | 24–30 | Very High | Moderate | Moderate | Low | Low | Bi-Monthly | Low |
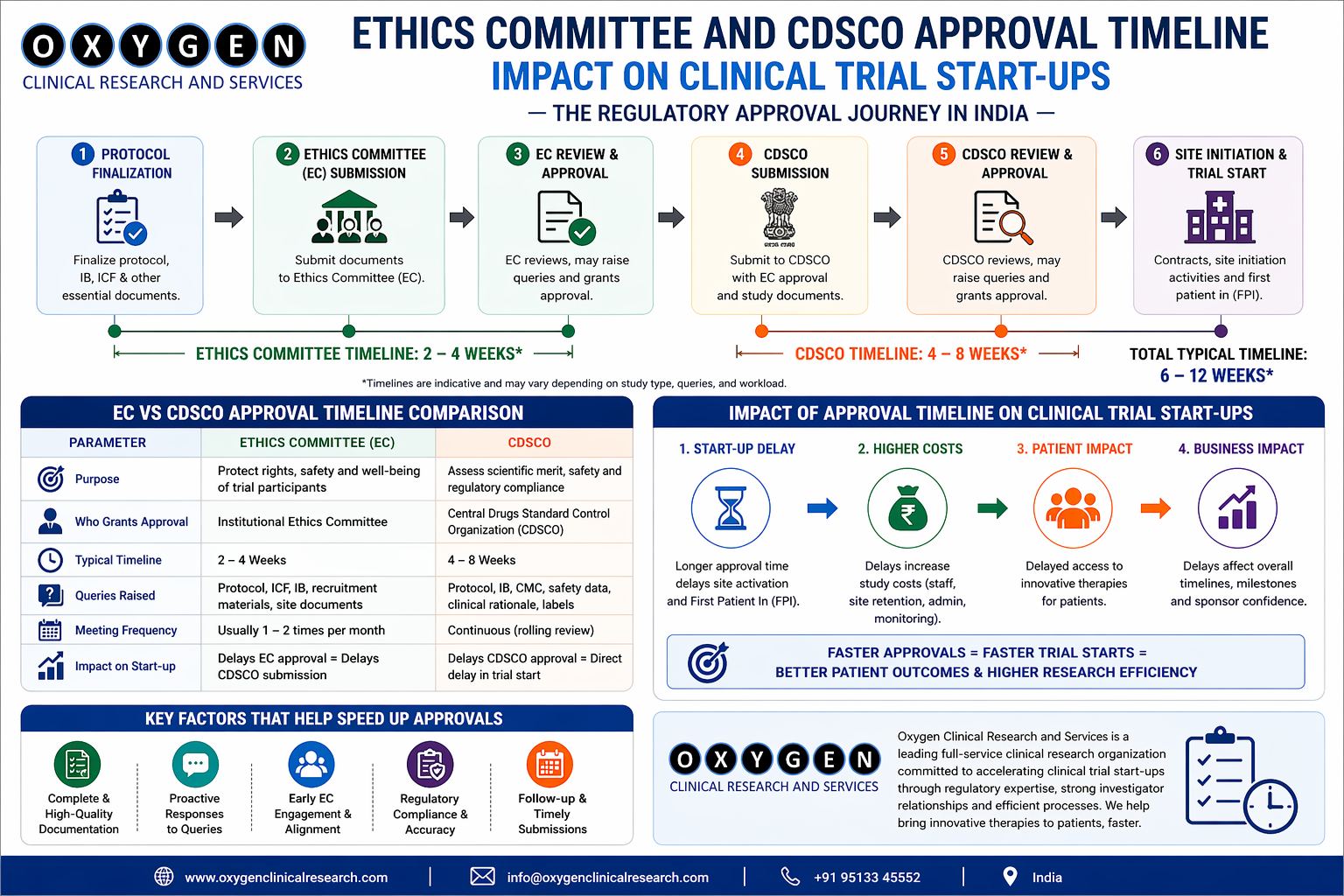
Indian Regulatory Approval Process and Selection Bottlenecks
Clinical Trial Site Selection The Indian regulatory landscape requires a dual-track mindset. While the CDSCO (Central Drugs Standard Control Organization) has streamlined the DCGI approval process to approximately 30–60 days for global studies, the local site-level bottlenecks remain. A site might have a brilliant PI, but if their Institutional Ethics Committee (IEC) meets once every three months and has an arduous 20-step submission process, your “fast-track” study will sit in a drawer. When we evaluate sites via Oxygen Clinical Research Services India, we look at the EC’s track record of queries. Are they asking relevant safety questions, or are they stalling on administrative trivialities?
The New Drugs and Clinical Trials Rules (2019) mandate specific compensation clauses and injury management protocols. Sites that lack a dedicated legal or administrative team frequently struggle to sign Clinical Trial Agreements (CTAs), leading to delays that can exceed three months.
Real Operational Insights: What Fails and Why
Most feasibility questionnaires are filled out by a junior study coordinator and signed by a PI who hasn’t read the protocol. To find the truth, look at these three indicators:
- Staff Stability: If the site has replaced three coordinators in the last year, your data quality will crash during the first monitoring visit.
- Competing Trials: Ask how many active trials the PI is currently leading. If they are the PI for four other Phase III trials in the same indication, your study is a low priority.
- Internal Logistical Chains: Does the site have an internal pharmacy, or do they outsource? Does the lab have a valid NABL accreditation? If these aren’t in place, your screening failures will skyrocket due to logistical errors, not patient eligibility.
Table 2: Operational Risk Assessment Matrix
| Sr. No. | Risk Factor | Probability | Impact on Timeline | Impact on Cost | Mitigation Strategy | Data Quality Effect | Monitoring Burden | Site Type Sensitivity |
| 1 | EC Delay | High | 3–5 Months | High | Select sites with monthly ECs | Minimal | High | Academic/Govt |
| 2 | Staff Turnover | Moderate | 1–2 Months | Moderate | Verify site-level SOPs for training | Severe | Very High | Corporate |
| 3 | PI Unavailability | High | Ongoing | Low | Appoint a strong Co-Investigator | High | High | Tier-1 Private |
| 4 | IP Storage Issues | Low | 1 Month | High | Temperature log audit during PSSV | Severe | Moderate | Small Clinics |
| 5 | Poor Recruitment | High | Indefinite | Severe | Patient database verification | Low | Moderate | All |
Case Studies: Real-World Execution Outcomes
Case Study 1: The “Paper Patient” Trap
- Study Type: Phase III Cardiovascular
- Site Type: Tier-1 Corporate Hospital
- Problem: Site promised 15 patients per month but delivered zero in the first quarter.
- Root Cause: The PI relied on referrals from other cardiologists in the hospital who were actually competitors and refused to refer patients to the trial.
- Action Taken: Moved the study to a private specialty clinic with a dedicated patient database managed by the PI.
- Outcome: Recruitment stabilized at 4 patients/month. Lost 4 months and approximately $50k in site management costs.
- Lesson Learned: Never trust referral-based recruitment models in India without written agreements from referring physicians.
Case Study 2: The EC Administrative Loop
- Study Type: Rare Disease Phase II
- Site Type: Government Academic Institute
- Problem: 8-month delay in study startup.
- Root Cause: The EC required the CTA to be signed before they would review the protocol, but the institute’s legal team would not sign the CTA until the EC granted approval.
- Action Taken: Direct intervention with the Dean to allow parallel processing.
- Outcome: Startup completed, but the delay forced the sponsor to rethink the entire South Asia strategy.
- Lesson Learned: Check the EC-Legal sequence protocol during feasibility, not after site selection.
Case Study 3: The Data Integrity Crisis
- Study Type: Phase III Dermatology
- Site Type: Small Private Research Center
- Problem: Major audit findings regarding source documentation.
- Root Cause: The PI used a single coordinator for three different studies, leading to “cloned” entries and missed vital sign windows.
- Action Taken: Terminated the site; transferred patients where possible; Clinical Trial Site Management India protocols were implemented to salvage data.
- Outcome: Data from this site was excluded from the final FDA submission; $80k loss.
- Lesson Learned: Assess the coordinator-to-study ratio. One coordinator cannot manage more than 2-3 intensive trials.
Challenges and Mitigation in Indian Sites
The biggest challenge is not the science; it is the infrastructure and “trial-readiness.” In India, you will face:
- Power Stability: Cold chain management is a frequent failure point. Sites must have 24/7 power backup with automated temperature logs. Manual logging is prone to fabrication.
- Calibration Records: Many sites forget to calibrate their weigh scales, thermometers, or BP apparatus. A monitor finds this six months in, and suddenly every piece of data collected is questionable.
- Informed Consent Logic: Audio-Video (AV) recording of the consent process is mandatory in certain trials in India. Sites often struggle with the storage and privacy hardware required for this.
Myths vs Reality
- Myth: India is “cheap” for clinical trials.
- Reality: While per-patient recruitment costs might be lower, the cost of high-quality monitoring and the heavy regulatory burden at the CDSCO level means the total cost of ownership is comparable to Eastern Europe if you want data that passes an FDA audit.
- Myth: Large hospitals always produce the best data.
- Reality: Large hospitals have the most bureaucracy and the least PI time. Private, dedicated research clinics often provide much higher data quality and faster response times.
Common Mistakes
Sponsor Mistakes
- Pushing for a “global” launch date in India without accounting for the mandatory CTRI (Clinical Trials Registry – India) registration which must be done before the first patient is enrolled.
- Ignoring the site’s previous history of regulatory queries.
CRO Mistakes
- Over-promising recruitment targets to win the bid.
- Assigning CRAs who are spread across 10 sites in 5 cities, leading to superficial monitoring.
Site Mistakes
- Treating clinical research as a side-revenue stream rather than a clinical commitment.
- Poor document retention practices that fail during long-term follow-up studies.
The Counterintuitive Insight: Avoid the “Star” PI
Most sponsors chase the “Key Opinion Leaders” (KOLs). In my experience, the KOL is your biggest risk factor. They are in Switzerland for a conference, they are at a national gala, or they are performing surgeries 12 hours a day. They have no time to check the Case Report Forms (CRFs).
Instead, select a site where the PI is mid-career, hungry for publication, and actively involved in the daily clinic. This PI will actually see the patient, and that is where data quality lives.
Practical Sponsor Checklist
Feasibility Stage
- Verify CTRI registration of previous studies.
- Check EC meeting frequency and average approval turnaround time.
- Count current active trials for the proposed PI.
- Confirm NABL accreditation for all local lab parameters.
Startup Stage
- Execute CTA with clear timelines for EC submission.
- Standardize the Informed Consent Form (ICF) to meet New Drugs and Clinical Trials Rules 2019.
- Verify AV recording hardware and privacy protocols.
Execution Stage
- Perform a pre-SIV (Site Initiation Visit) check of pharmacy and storage.
- Audit the training logs of the Study Coordinator.
- Implement a 24-hour query resolution requirement for the site.
Regulatory and Compliance Context
Navigating the Indian environment requires strict adherence to:
- CDSCO / DCGI: The primary regulatory gatekeeper.
- ICMR (Indian Council of Medical Research): Ethical guidelines that often go beyond ICH-GCP.
- CTRI: Mandatory registration before the first patient is enrolled.
- ICH-GCP E6(R3): The global standard that Indian sites must meet for international acceptance.
For those looking to establish a footprint, understanding the Clinical Research Contact India requirements is the first step in avoiding early administrative rejection.
Suggested Infographics (Concept)
- The India Approval Funnel: A diagram showing the timeline from CDSCO submission to DCGI approval, highlighting the parallel EC and CTA execution.
- Site Risk Heatmap: A visual comparison of Corporate vs. Private vs. Academic sites across Recruitment, Quality, and Speed.
- The Enrollment Gap: A chart showing the difference between “Feasibility Promises” and “Actual Accrual” based on historical data.
External References
- CDSCO (Central Drugs Standard Control Organization)
- ICMR Ethical Guidelines
- CTRI (Clinical Trials Registry – India)
- New Drugs and Clinical Trials Rules, 2019.
FAQ Section
1. How long does the average site selection and startup take in India? From initial feasibility to Site Initiation Visit (SIV), you should budget 4 to 6 months. While regulatory approval is faster now, the site-level hurdles—EC approvals and CTA negotiations—remain the primary delay factors.
2. Is patient recruitment in India still as fast as it used to be? Yes, but the quality has changed. Regulatory oversight is much stricter. You can still recruit quickly, but you must ensure that every patient is truly eligible and that the consent process is perfectly documented to survive an audit.
3. What is the biggest error made during feasibility? Relying on “database numbers.” Sites see thousands of patients, but only a fraction meet the restrictive inclusion/exclusion criteria of a modern protocol. You must perform an actual “chart review” during feasibility.
4. Are local ethics committees reliable for global trials? Many private and corporate hospitals have highly efficient and ICH-GCP compliant ECs. However, some smaller or older academic centers have committees that are understaffed and slow. Always audit the EC’s SOPs before selecting a site.
5. How has the 2019 Rule changed site selection? It has moved the focus toward legal and administrative compliance. Sites must now be much more aware of their responsibility regarding patient compensation and reporting of Serious Adverse Events (SAEs) within 24 hours.
Execution in India is about managing the specifics of each site rather than hoping for a generic outcome. Success requires a partner who understands the local culture, the regulatory nuances, and the difference between a site that looks good on paper and one that delivers quality data. For strategic execution, you can reach out via Oxygen Clinical Trial or connect with me directly Clinical Trial Site Selection.
Contact Information: Email: govindpawar@oxygenclinicaltrials.com LinkedIn: Govind Pawar Profile