Oxygen Clinical Research: Ensuring Safe and Reliable Clinical Trials
The pursuit of groundbreaking medical advancements hinges on the successful execution of clinical trials. These meticulously designed studies are the bridge between promising scientific discoveries and life-altering therapies available to patients. At the core of this critical endeavor lies the paramount importance of safety and reliability. Oxygen Clinical Research recognizes this fundamental principle and has built its operations around a robust framework designed to ensure that every trial it undertakes is conducted with the utmost integrity, prioritizing participant well-being and the generation of accurate, trustworthy data.
Oxygen Clinical Research’s commitment to safety and reliability is not a mere afterthought; it is deeply embedded within its organizational culture and permeates every stage of the clinical trial lifecycle. This dedication is evident in their multifaceted approach, which encompasses stringent protocol adherence, comprehensive site selection and monitoring, rigorous data management and quality control, and a proactive approach to risk assessment and mitigation.
I. Unwavering Adherence to Protocol and Regulatory Compliance:
The cornerstone of any safe and reliable clinical trial is the strict adherence to the approved protocol. Oxygen Clinical Research meticulously develops and implements protocols in strict accordance with international ethical and scientific quality guidelines, including the Declaration of Helsinki, Good Clinical Practice (GCP) guidelines, and relevant regulatory authority requirements (e.g., FDA, EMA).
- Protocol Development and Review: Each protocol is meticulously crafted by experienced medical professionals and researchers, incorporating the latest scientific knowledge and ethical considerations. These protocols undergo rigorous internal review and, where applicable, external expert consultation to ensure scientific validity, patient safety, and feasibility.
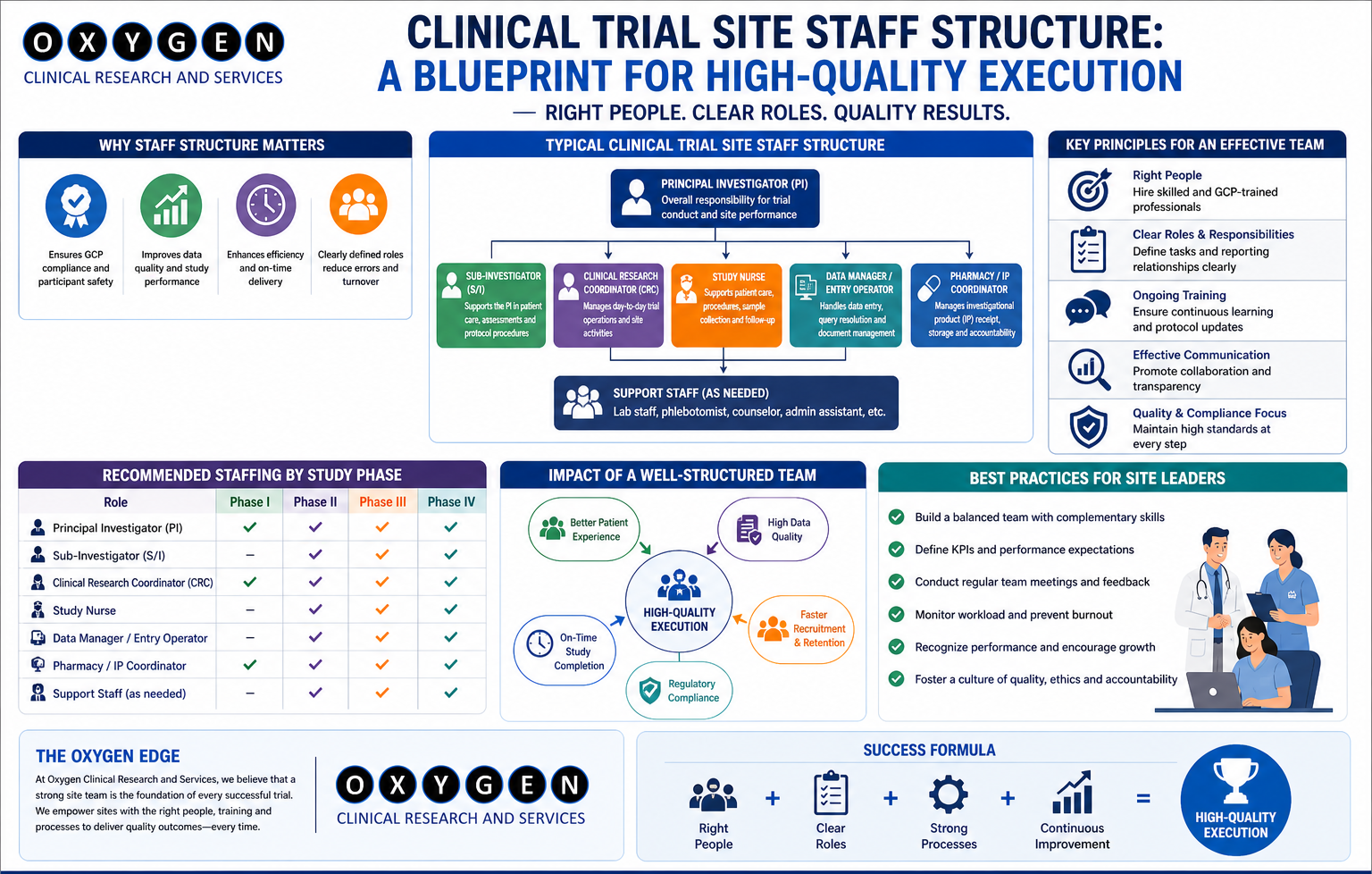
- Investigator Training and Qualification: Oxygen Clinical Research invests heavily in ensuring that all investigators and their study teams are thoroughly trained on the specific protocol, study procedures, and relevant safety monitoring requirements. This includes comprehensive training on Good Clinical Practice (GCP), informed consent procedures, adverse event reporting, and proper data collection techniques. Investigators are carefully selected based on their expertise, experience, and the availability of adequate resources.
- Informed Consent Process: The informed consent process is a critical element of participant safety and autonomy. Oxygen Clinical Research ensures that the informed consent form is written in clear, understandable language, fully detailing the study’s purpose, procedures, potential risks and benefits, alternatives, and the voluntary nature of participation. They emphasize a thorough and unhurried discussion between the investigator and the potential participant to ensure complete comprehension and address all questions before enrollment.
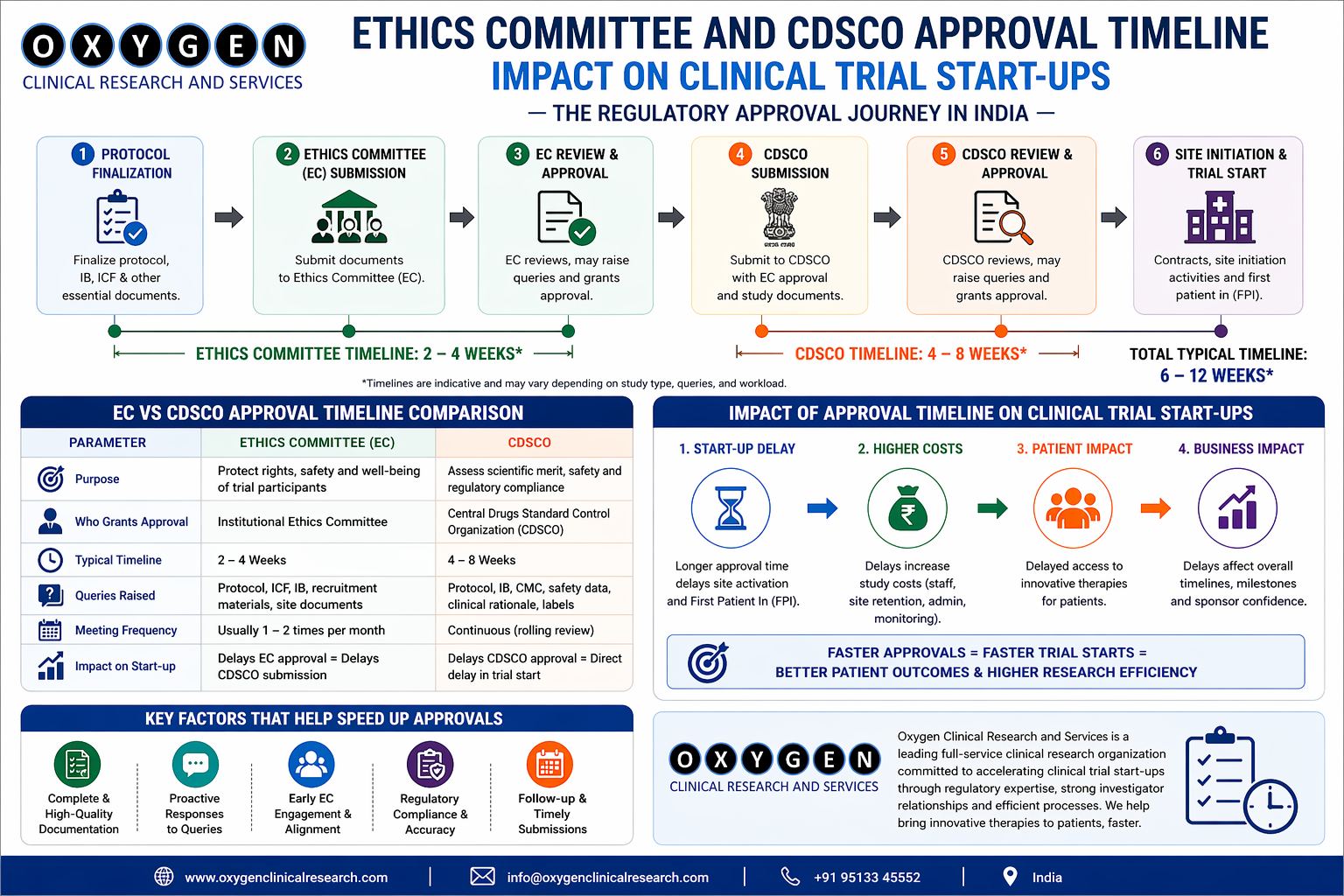
- Regulatory Adherence: Oxygen Clinical Research maintains a proactive stance on regulatory compliance. Their teams are continuously updated on evolving regulatory landscapes and ensure that all submissions, amendments, and reporting activities are timely and accurate, fostering a transparent relationship with regulatory bodies.
II. Paramount Site Selection and Robust Monitoring: The quality of a clinical trial is intrinsically linked to the performance of the research sites. Oxygen Clinical Research employs a rigorous site selection process and implements comprehensive monitoring strategies to ensure adherence to protocols and maintain participant safety.
- Site Qualification and Selection: Potential research sites are meticulously evaluated for their infrastructure, investigator and staff expertise, patient recruitment capabilities, experience with similar studies, and commitment to GCP principles. Site qualification visits are conducted by experienced clinical research associates (CRAs) to assess the site’s readiness and suitability.
- Proactive Monitoring and Oversight: Oxygen Clinical Research’s monitoring strategy is not merely reactive; it is proactive and data-driven. CRAs conduct regular site visits (both remote and on-site) to verify data accuracy, ensure adherence to the protocol and regulatory requirements, review source documentation, assess patient safety, and provide ongoing support and training to site staff. This includes source data verification (SDV), review of regulatory binders, and assessment of investigational product handling.
- Risk-Based Monitoring (RBM): Recognizing that not all trial data and processes carry equal risk to patient safety or data integrity, Oxygen Clinical Research increasingly employs a Risk-Based Monitoring (RBM) approach. This strategy focuses monitoring efforts on critical data and processes identified as having the greatest potential to impact patient safety or data reliability, thereby optimizing resource allocation and enhancing early detection of potential issues.
III. Rigorous Data Management and Quality Control: Accurate and reliable data is the bedrock of meaningful clinical trial results. Oxygen Clinical Research employs sophisticated data management systems and stringent quality control measures to ensure data integrity.
- Electronic Data Capture (EDC) Systems: Oxygen Clinical Research utilizes advanced EDC systems for efficient and accurate data collection. These systems incorporate built-in edit checks and data validation rules to minimize errors at the point of entry, ensuring data quality from the outset.
- Data Validation and Cleaning: A multi-layered data validation and cleaning process is implemented. This includes automated checks within the EDC system, manual review of data queries by trained data managers, and statistical review for anomalies.
- Centralized Data Monitoring: Data trends and potential data quality issues are continuously monitored centrally by dedicated data management teams. This allows for prompt identification of systemic errors or site-specific issues that may require immediate attention.
- Quality Assurance (QA) Audits: Independent QA audits are conducted regularly to assess the compliance of trial operations with protocols, GCP, and regulatory requirements. These audits provide an objective evaluation of the quality management system and identify areas for improvement.
IV. Proactive Risk Assessment and Mitigation: Anticipating and addressing potential risks is crucial for maintaining both safety and reliability. Oxygen Clinical Research adopts a forward-thinking approach to risk management.
- Identification of Potential Risks: From the initial protocol design phase through study execution, potential risks to patient safety, data integrity, and operational efficiency are systematically identified. This includes risks associated with investigational product handling, patient recruitment challenges, protocol deviations, and data management issues.
- Development of Mitigation Strategies: For each identified risk, specific mitigation strategies are developed and implemented. This might involve enhanced monitoring plans, additional training for specific site personnel, development of standardized operating procedures (SOPs), or the implementation of specific data management checks.
- Adverse Event (AE) and Serious Adverse Event (SAE) Management: Oxygen Clinical Research has robust systems in place for the prompt and accurate reporting, assessment, and reconciliation of AEs and SAEs. All AEs are meticulously documented, evaluated for causality and severity, and reported to the sponsor and regulatory authorities in accordance with established timelines.
- Continuous Improvement: The lessons learned from risk assessments, monitoring activities, and audits are fed back into the organizational learning process, driving continuous improvement in all aspects of clinical trial conduct.
conclusion: Oxygen Clinical Research’s commitment to ensuring safe and reliable clinical trials is a testament to their comprehensive and proactive approach. By prioritizing strict protocol adherence, meticulous site selection and monitoring, rigorous data management, and proactive risk mitigation, Oxygen Clinical Research consistently delivers high-quality clinical trial data that is both scientifically sound and ethically conducted. This unwavering dedication to safety and reliability not only protects the well-being of study participants but also provides the pharmaceutical and biotechnology industries with the trustworthy evidence needed to advance medical innovation and bring life-changing therapies to patients worldwide.