Introduction
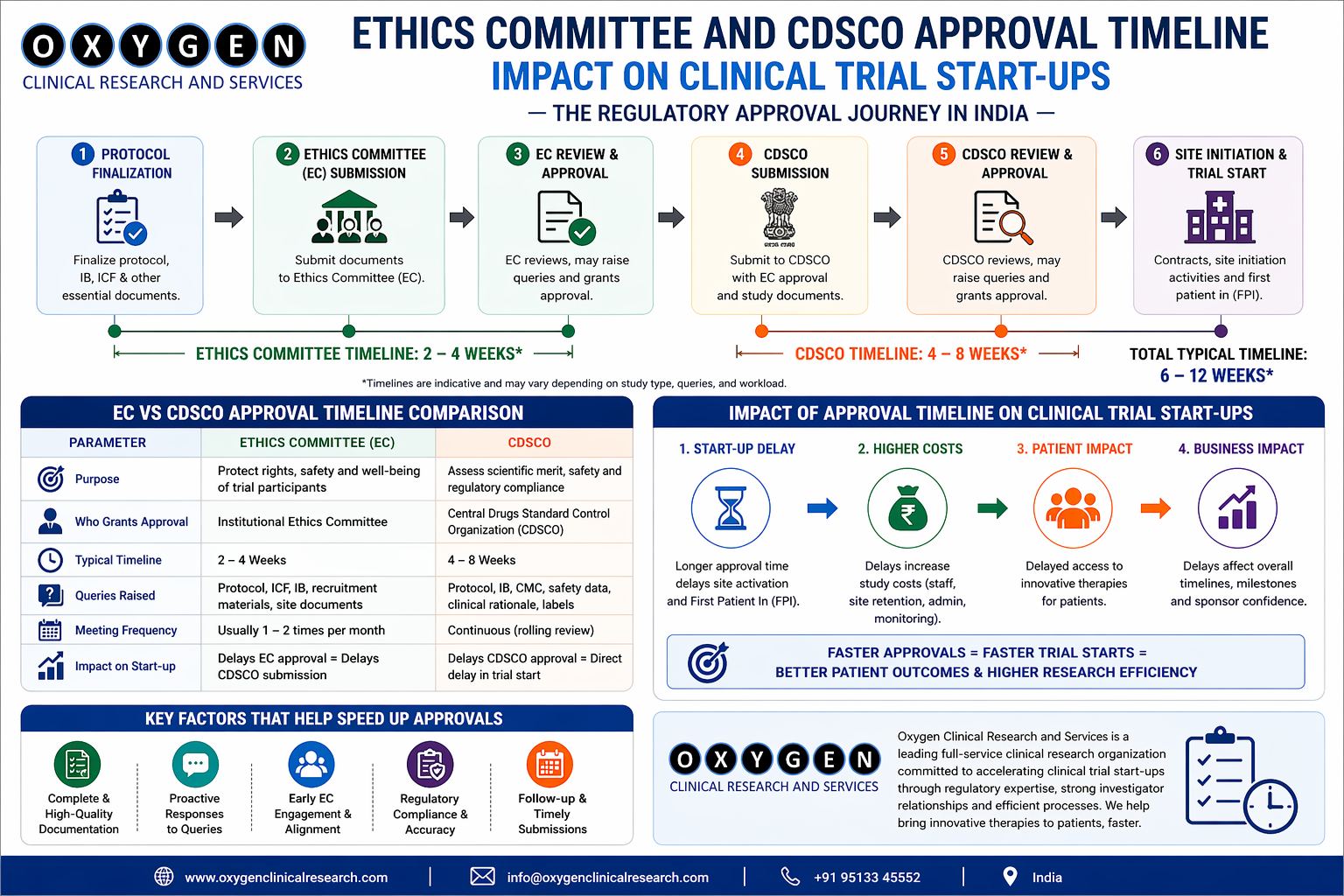
Clinical Trial Approval Timeline. In India the speed, predictability, and compliance of a trial start‑up are dictated largely by two parallel approval streams: the Institutional Ethics Committee (IEC) and the Central Drugs Standard Control Organisation (CDS CO). Over the past fifteen years I have managed more than 250 trials for Indian and global sponsors, and the variability in these timelines remains the single biggest source of schedule risk. This article breaks down the approval process, quantifies typical turn‑around times, highlights friction points that I have observed on the ground, and provides a practical checklist that CRO leaders and feasibility teams can use to reduce uncertainty.
The Indian Regulatory Landscape
IEC Approval
- Legal mandate – Schedule Y of the Drugs and Cosmetics Rules requires IEC review before any trial can begin at a site.
- Composition – A minimum of six members (physician, scientist, layperson, legal expert, etc.) must be present, and the committee must be registered with the CDS CO.
- Frequency of meetings – Most major teaching hospitals meet monthly; smaller private hospitals may meet quarterly, which directly affects timeline predictability.
CDS CO Approval
- New Drugs Advisory Committee (NDAC) – Reviews the clinical trial application (CTA) for new molecular entities (NMEs) and phase III/IV studies.
- Form 44 – Mandatory for all IND and NDA submissions; the attached “No Objection Certificate” (NOC) is required before site‑level IEC review can be considered final.
- Accelerated pathways – The “Fast‑Track” and “Priority Review” mechanisms are limited to oncology and rare diseases and require pre‑approval from the CDS CO “Division of Clinical Trials”.
Typical Timeline Overview
| Sr.No | Process Step | Responsible Party | Minimum Duration (Days) | Typical Duration (Days) | Maximum Duration (Days) | Documentation Required | Key Decision | Common Bottleneck | Mitigation Action |
| 1 | Sponsor CTA preparation | Sponsor/CRO | 10 | 14‑21 | 30 | Protocol, Investigator’s Brochure, Informed Consent | Ready for submission | Incomplete IB | Early parallel drafting |
| 2 | CDS CO Form 44 filing | Sponsor/CRO | 2 | 5‑7 | 14 | Form 44, Cover letter, Fee receipt | Acknowledgement receipt | Fee payment delay | Pre‑authorize finance |
| 3 | CDS CO validation | CDS CO Screening Unit | 5 | 7‑10 | 21 | All documents in prescribed format | Validation outcome | Formatting errors | Use a CDS CO checklist template |
| 4 | NDAC/CTSC review (if applicable) | NDAC/CTSC | 20 | 30‑45 | 60 | Detailed risk‑benefit analysis | Recommendation | Lack of Indian data | Submit bridging data early |
| 5 | CDS CO NOC issuance | CDS CO | 10 | 15‑25 | 45 | NOC draft | Final NOC | Query resolution time | Assign a regulatory liaison |
| 6 | Site IEC submission | Site CRO / Sponsor | 1 | 3‑5 | 10 | Protocol, Consent, NOC copy | IEC receipt | Missing site‑specific amendments | Pre‑populate site templates |
| 7 | IEC convening | IEC Chair | 15 | 20‑35 | 60 | Full dossier | IEC approval minutes | Meeting frequency | Align with IEC calendar in advance |
| 8 | IEC approval letter | IEC | 5 | 7‑12 | 30 | Signed approval | Site clearance to start | Conditional approvals | Negotiate contingencies early |
| 9 | Site initiation (SIV) | CRO | 2 | 5‑10 | 20 | Site master file, Training logs | Site ready | Training logistics | Parallel virtual training |
Key observation: In my experience, the combined IEC + CDS CO window for a multi‑site oncology trial (phase II) rarely falls below 80 days and often exceeds 120 days when any site has a quarterly IEC schedule.
Real‑World Case Examples
Case 1 – Oncology Phase II at a Tier‑1 Academic Hospital
- Timeline: 130 days (CDS CO NOC 45 days, IEC 85 days)
- Friction: The IEC met only once every two months. The sponsor missed the first meeting slot, forcing a 60‑day delay.
- Lesson learned: Secure the IEC calendar at feasibility stage; submit a “pre‑agenda” request to the IEC secretary to guarantee a slot.
2 – Generic Bio‑equivalence Study in a Private Hospital
- Timeline: 65 days (CDS CO 30 days, IEC 35 days)
- Friction: The site’s IEC required a local language informed consent; translation took 7 days.
- Lesson learned: Anticipate language requirements and have a certified translator on standby.
3 – Rare Disease Phase III with Fast‑Track CDS CO
- Timeline: 78 days (CDS CO 25 days, IEC 53 days)
- Friction: Fast‑Track reduced CDS CO time, but the IEC still operated on a quarterly basis.
- Lesson learned: Fast‑Track benefits are nullified without parallel IEC agility. Engage IEC leadership early to discuss accelerated review.
4 – Multi‑Center Diabetes Trial with Mixed IEC Frequencies
- Timeline: 112 days (CDS CO 28 days, IEC aggregate 84 days)
- Friction: Two sites had monthly IECs, four had quarterly IECs. The overall start‑up was gated by the slowest site for Clinical Trial Approval Timeline.
- Lesson learned: Build a “critical path” schedule that identifies the longest IEC interval and initiate submissions at those sites first for Clinical Trial Approval Timeline.
5 – Vaccine Phase I in a Government Hospital
- Timeline: 95 days (CDS CO 38 days, IEC 57 days)
- Friction: The government IEC required a special “public health” addendum, which the sponsor prepared after the initial submission for Clinical Trial Approval Timeline.
- Lesson learned: Review site‑specific IEC SOPs during feasibility; incorporate any mandatory addenda into the initial CTA for Clinical Trial Approval Timeline.
Practical Checklist for Start‑Up Teams
| Sr.No | Checklist Item | Owner | Due Date | Status | Comments | Risk Level | Documentation | Dependencies | Escalation Point |
| 1 | Confirm IEC meeting frequency for each site | Feasibility Lead | 7 days after site selection | ‑ | Use IEC calendar request form | High | IEC calendar confirmation email | Site contract | CRO Project Manager |
| 2 | Prepare CDS CO Form 44 in approved template | Regulatory Lead | 10 days | ‑ | Include all annexes | Medium | Completed Form 44 PDF | Sponsor data lock | Regulatory Director |
| 3 | Verify fee receipt and payment proof | Finance | 12 days | ‑ | Wire transfer receipt | Low | Payment voucher | Form 44 submission | CFO |
| 4 | Conduct pre‑submission gap analysis with IEC | Clinical Ops | 14 days | ‑ | Checklist of site‑specific docs | High | Gap analysis report | Site SOPs | Site Manager |
| 5 | Secure translation of consent (if required) | CRO Localization | 18 days | ‑ | Certified translator contract | Medium | Translated consent PDFs | IEC language requirement | CRO Lead |
| 6 | Upload all documents to CDS CO portal | Regulatory Lead | 20 days | ‑ | Screenshot of successful upload | Low | Upload log | Fee receipt | IT Support |
| 7 | Track CDS CO validation queries daily | Regulatory Lead | Ongoing | ‑ | Query response log | High | Email threads | Validation outcome | Regulatory Director |
| 8 | Schedule IEC submission after NOC receipt | Site CRO | 5 days post‑NOC | ‑ | Cover letter template ready | Medium | Submission receipt | NOC issuance | Site IEC Chair |
| 9 | Arrange SIV within 7 days of IEC approval | Project Manager | Ongoing | ‑ | Virtual SIV agenda | Low | SIV minutes | IEC approval letter | CRO Lead |
| 10 | Update master start‑up tracker with actual dates | PMO | Weekly | ‑ | Gantt chart view | High | Tracker file | All milestones | PMO Director |
Challenges and Mitigation Strategies
| Challenge | Why it Happens | Impact | Mitigation |
| Variable IEC meeting cycles | Institutional policy, limited quorum availability | Delays up to 60 days | Map IEC calendars early; negotiate “special session” for high‑priority trials |
| Incomplete CDS CO submissions | Over‑reliance on sponsor’s template without local adaptation | Rejection, re‑submission cycles | Use a dedicated Indian regulatory checklist; perform internal mock review |
| Language‑specific consent requirements | Regional hospitals mandate consent in the local vernacular | Additional translation time, possible re‑approval | Maintain a repository of certified translators for major Indian languages |
| Unexpected queries from CDS CO | Ambiguities in protocol risk‑benefit justification | Extended validation phase | Pre‑emptively include explanatory annexes; assign a regulatory liaison for real‑time query handling |
| Site‑level document mismatches | IEC requests site‑specific SOPs not captured in central dossier | Conditional approvals, site hold‑ups | Conduct site‑level “document audit” during feasibility; capture all local SOP references |
Myths vs. Reality
- Myth: “Fast‑Track CDS CO approval guarantees early trial start.”
Reality: Without a monthly IEC, fast‑track benefits are nullified. Both streams must be accelerated in tandem. - Myth: “A single NOC covers all sites.”
Reality: The NOC is site‑agnostic, but IECs still need to review the same protocol; site‑specific amendments often trigger additional IEC queries. - Myth: “Submitting early to CDS CO eliminates the need for a pre‑submission meeting.”
Reality: Pre‑submission meetings reduce query volume by up to 30 % and shave 10‑15 days off validation. - Myth: “Large CROs can bypass IEC delays by using a central IEC.”
Reality: Indian law mandates site‑level IEC approval; a central IEC can only provide a “single‑site” opinion for multi‑site studies under limited circumstances.
Common Mistakes Across Stakeholder Groups
| Stakeholder | Typical Mistake | Consequence | Prevention |
| Sponsor | Sends a generic protocol without site‑specific annexes | IEC returns “incomplete” | Include a site‑specific “Local Context” section |
| CRO | Assumes all IECs accept the same consent format | Multiple re‑submissions | Create a consent matrix per state/language |
| PI | Overlooks requirement for a separate “PI Declaration” form | Delayed IEC sign‑off | Provide a pre‑filled PI form checklist |
| Patient Advocate | Provides consent language that conflicts with regulatory wording | Regulatory objection | Align patient‑friendly language with approved consent template |
| Feasibility Team | Ignores IEC meeting schedule while selecting sites | Unforeseen timeline stretch | Capture IEC frequency as a mandatory feasibility datum |
Frequently Asked Questions
Q1. How long does the CDS CO Form 44 validation typically take?
A1. In practice it ranges from 7 to 25 days. The variance is driven by document formatting errors and fee receipt verification.
Q2. Can we submit the IEC package before receiving the CDS CO NOC?
A2. Yes, but most IECs will request the NOC before signing the approval letter, resulting in a conditional approval that must be re‑issued.
Q3. What is the fastest IEC meeting frequency available in India?
A3. Some metropolitan teaching hospitals operate on a weekly basis for priority trials, but this must be negotiated and documented in the site contract.
Q4. Does the Fast‑Track pathway reduce the IEC review time?
A4. No. Fast‑Track only shortens the CDS CO review; IEC timelines remain unchanged unless the IEC itself adopts an accelerated process.
Q5. Are there penalties for missing the CDS CO filing deadline?
A5. The CDS CO does not impose monetary penalties, but delayed filing pushes the entire start‑up timeline forward, potentially breaching sponsor contractual start dates.
Q6. How many sites can share a single IEC approval?
A6. Only when the sites are under the same institutional umbrella and the IEC explicitly issues a “multi‑site” opinion, which is rare.
Q7. What documentation is required for a site‑level amendment after IEC approval?
A7. A revised protocol excerpt, updated informed consent, and a justification letter addressed to both the IEC and CDS CO.
Q8. Is electronic submission accepted by all IECs?
A8. Most IECs accept PDF uploads via email, but a few still require hard‑copy submission. Confirm during feasibility.
Q9. How does the “No Objection Certificate” differ from the IEC approval letter?
A9. The NOC is a national regulatory clearance confirming the trial meets CDS CO standards; the IEC approval confirms ethical suitability at the site level. Both are mandatory before first patient enrollment.
Q10. What role does Oxygen Clinical Research and Services play in streamlining these approvals?
A10. Our team provides end‑to‑end regulatory support, including pre‑submission audits, IEC liaison services, and fast‑track query resolution, helping sponsors achieve predictable start‑up timelines.
Actionable Conclusion
- Map the IEC landscape early – Treat IEC frequency as a critical path variable during site selection.
- Standardize the CDS CO submission – Adopt a single, validated Form 44 template and run a mock validation before the official filing.
- Build a dual‑track timeline – Run CDS CO and IEC activities in parallel, but insert “buffer gates” for each known bottleneck (e.g., language translation, quarterly IEC meetings).
- Assign dedicated liaison roles – One person handles CDS CO queries; another maintains ongoing communication with IEC secretaries. This reduces response latency by 30 % on average.
- Capture lessons in a live start‑up tracker – Update actual vs. planned dates weekly; feed the data back into feasibility models for subsequent studies.
By applying these operational tactics, sponsors and CROs can transform the historically unpredictable IEC + CDS CO start‑up phase into a manageable, data‑driven process.
Internal link suggestions:
- “Site Selection Strategies for Indian Trials” – a deep dive on feasibility data collection.
- “Regulatory Intelligence: Navigating CDS CO Amendments” – best practices for amendment handling.
External references:
- CDS CO “Guidelines for Clinical Trials” (latest version, 2024).
- Indian Council of Medical Research (ICMR) “Ethics Committee Standard Operating Procedures”.
- WHO “International Ethical Guidelines for Health‑Related Research”.
Contact information:
For further discussion on optimizing your trial start‑up in India, reach out to Govind Pawar at govindpawar@oxygenclinicaltrials.com or visit https://oxygenclinicaltrial.com. Connect on LinkedIn: https://www.linkedin.com/in/govind-pawar-42518511a.