Clinical Trial SQV Process. I have spent fifteen years standing in hospital corridors, sitting across from overworked Principal Investigators (PIs), and reviewing site files that looked perfect on paper but were a liability in reality. I have seen trials delayed by six months because a site was qualified based on “reputation” rather than operational reality. I have seen data get tossed during a CDSCO audit because a site’s source documentation didn’t meet ALCOA+ standards, despite a “successful” SQV. A Site Qualification Visit (SQV) is frequently treated as a box-ticking exercise by junior monitors or rushed CROs. This is a mistake that costs sponsors millions. In India’s unique regulatory and clinical landscape, the SQV is the only barrier between a successful trial and a multi-year regulatory nightmare. If you don’t find the cracks during the SQV, you will find them during the first patient audit.
Executive Summary: The Sponsor Perspective on Risk
For a Sponsor or a Clinical Operations head, the SQV is about three things: predictability, compliance, and ROI. Every day a site is stagnant costs thousands of dollars. In India, the “New Drugs and Clinical Trials Rules (2019)” have streamlined some aspects, but the complexity of Ethics Committee (EC) management and investigator commitment remains a bottleneck Clinical Trial SQV Process.
What is at stake during an SQV?
- Timeline Integrity: Can the site actually recruit the promised 20 patients in 4 months?
- Financial Risk: Is the site overhead and infrastructure worth the investment?
- Regulatory Security: Is the PI’s team trained on CDSCO and ICH-GCP E6(R3) guidelines?
If a site fails at the SQV stage, it is a win. The real failure is qualifying a site that should never have been selected in the first place.
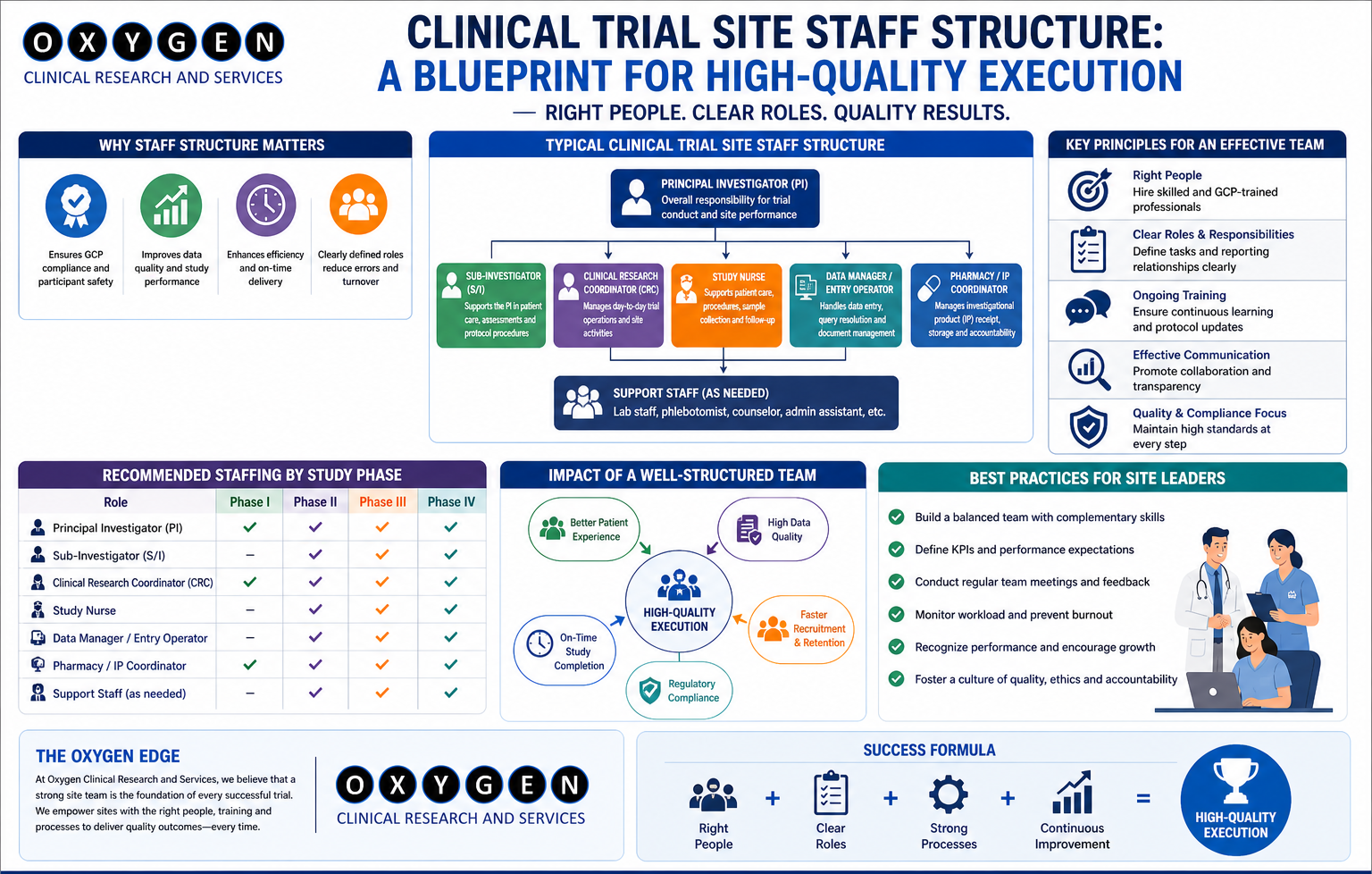
The Site Qualification Process: Beyond the Facility Tour
When we walk into a site for an SQV in India, we are looking for a culture of compliance. Most PIs will show you their high-end diagnostic equipment or their CV. As a seasoned leader, I look at the study coordinator’s desk and the site’s Standard Operating Procedures (SOPs) Clinical Trial SQV Process.
The PI Interview: Assessing Real Commitment
Many Indian PIs are “academic giants” but “operational ghosts.” During the SQV, I evaluate if the PI is actually going to oversee the trial or if they are delegating everything to a junior resident.
- Does the PI understand the primary endpoint?
- Does the PI know the specific exclusion criteria that usually cause screening failures in the Indian population?
- How many other trials are they currently running? If they have 15 ongoing trials, your study will likely be neglected.
Staffing and Infrastructure
We evaluate the Site Management Organization (SMO) support if applicable. If a site relies on a single coordinator for three different therapeutic areas, your data quality will suffer. We check for:
- Equipment Calibration: Are the centrifuges and -80°C freezers calibrated? In India’s climate, power backups for cold chain storage are non-negotiable.
- Space and Security: Is there a locked, fire-secure area for the Trial Master File (TMF)?
- Pharmacy: How is the Investigational Product (IP) handled? Does the pharmacist understand temperature excursion reporting?
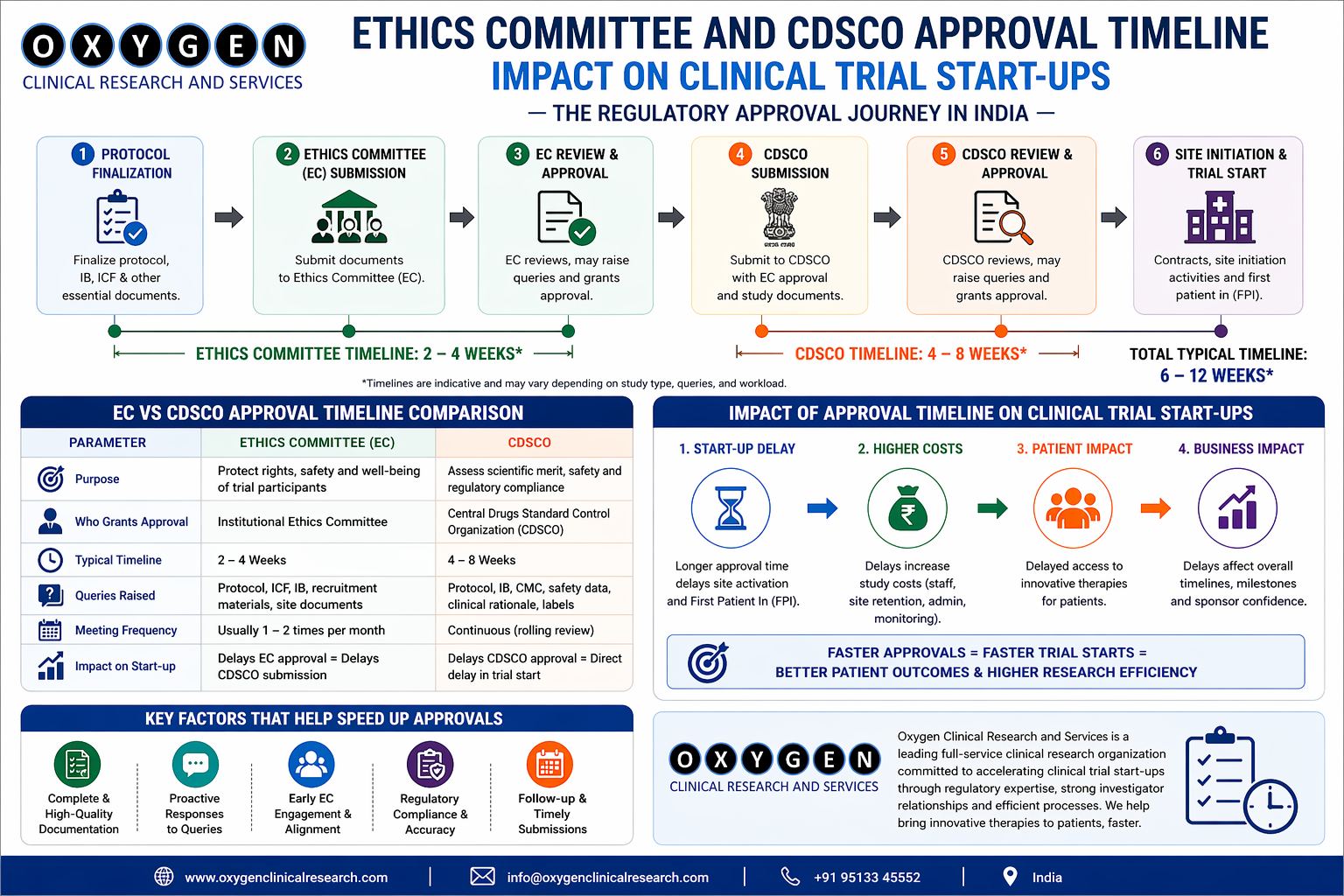
Where the Delays Happen: The Indian Context
Indian clinical trials often stall at the transition between SQV and Site Initiation Visit (SIV). The SQV must identify these potential “silent delays”:
- Ethics Committee (EC) Timelines: Many institutional ECs in India meet only once a month or once every two clouds. If you miss the submission deadline by one day, you lose at least 30 days. We verify the EC’s CDSCO registration status during the SQV. An unregistered EC is a non-starter.
- Contracting: Does the hospital’s legal department take three months to review a simple Clinical Trial Agreement (CTA)?
- CTRI Registration: The delay in Clinical Trials Registry – India (CTRI) registration often stems from poor documentation provided by the site during the qualification phase.
Learn more about navigating clinical research services in India
Real Operational Insights: What Actually Works vs. What Fails
Sites often fail not because they lack equipment, but because they lack process. In my experience, a Tier-2 city hospital with a dedicated PI and a meticulous coordinator often outperforms a “prestigious” metro hospital where the PI is rarely on-site.
Evidence of Recruitment Capability
During an SQV, don’t just ask for a recruitment estimate. Ask to see the de-identified patient logs from the last six months. If a site says they see 50 diabetic patients a week but cannot show a log to prove it, they are guessing. Guessing leads to recruitment failure.
ALCOA+ Compliance
Data must be Attributable, Legible, Contemporaneous, Original, Accurate, and now Complete, Consistent, Enduring, and Available. I have seen sites using scrap paper for primary vitals then “transcribing” them into source notes. This is a red flag. During the SQV, we must verify that the site has a system for capturing data at the point of care.
Case Studies: Lessons from the Field
Case Study 1: The Celebrity PI Trap
- Study Type: Phase III Cardiovascular Trial
- Site Type: Large Private Hospital, Metro City
- Problem: Enrollment was zero for three months after SIV.
- Root Cause: The PI was essentially a brand ambassador for the hospital and had no interest in the protocol. The study coordinator was a temp with no GCP training.
- Action Taken: Site was closed early; resources moved to a regional center.
- Outcome: Lost $15,000 in startup costs and 4 months of time.
- Lesson Learned: Never qualify a site based on the PI’s name alone. Evaluate the “Sub-Investigators” who will actually do the work.
Case Study 2: The Cold Chain Crisis
- Study Type: Vaccine Trial
- Site Type: Government Medical College
- Problem: IP temperature excursion during weekend power outage.
- Root Cause: The SQV missed that the site’s backup generator was not connected to the pharmacy’s dedicated refrigerator.
- Action Taken: Re-training and installation of a wireless temperature monitoring system with SMS alerts.
- Outcome: 200 doses of IP destroyed; $12,000 loss and a regulatory query from DCGI.
- Lesson Learned: Check the physical wiring of the backup power during the SQV. Don’t take “we have a generator” as an answer.
Case Study 3: The Unregistered Ethics Committee
- Study Type: Oncology Observational Study
- Site Type: Specialized Cancer Center
- Problem: Clinical trial data was declared invalid by the sponsor’s QA.
- Root Cause: The SQV auditor failed to check if the Institutional Ethics Committee (IEC) had renewed its CDSCO registration.
- Action Taken: Retrospective approval sought (which failed); trial restarted at a new site.
- Outcome: Loss of 6 months of data and a significant compliance hit.
- Lesson Learned: Always verify the IEC’s CDSCO and ICMR registration numbers and their validity dates before site selection.
Case Study 4: The Recruitment Overestimate
- Study Type: Rare Disease Hybrid Trial
- Site Type: Multi-specialty Hospital
- Problem: The site promised 10 patients but delivered only 1.
- Root Cause: The PI’s patient pool was largely “floating” (non-local) patients who could not return for the required 12 follow-up visits.
- Action Taken: Increased travel reimbursement for patients to maintain retention.
- Outcome: Increased trial cost by 20% to save the data point.
- Lesson Learned: Evaluate patient “stickiness” and geographic distance from the site during qualification.
Challenges and Mitigation: The Unfiltered Reality
Executing trials in India involves navigating a mix of high-tech facilities and legacy bureaucratic hurdles.
- High Staff Turnover: The person you train during the SQV is often gone by the time the first patient is screened.
Mitigation: Ensure the site has a documented internal training SOP. Don’t just train the coordinator; train the site.
- Regulatory Fluidity: CDSCO guidelines change. The New Drugs and Clinical Trials Rules (2019) introduced specific timelines for compensation in case of trial-related injury.
Mitigation: Ask the PI how they handle SAE (Serious Adverse Event) reporting. If their response is “the CRO handles it,” they are a liability.
- Diagnostic Discrepancies: A site might have a lab, but is it NABL-accredited? If you rely on a local lab for primary endpoints, you risk data rejection by the FDA or EMA.
Mitigation: Verify NABL/CAP certifications during the SQV or plan for a central lab. Our site management expertise addresses these specific risks
Myths vs. Reality
- Myth: Large government hospitals are too slow for commercial trials.
Reality: While the bureaucracy is real, government hospitals in India often have the highest patient volumes and the most loyal patient pools for long-term follow-up studies.
- Myth: A “GCP Certificate” means the staff is ready.
Reality: A certificate is a piece of paper. During an SQV, I ask the staff to explain the Informed Consent Form (ICF) process for an illiterate patient. Their answer tells me more than any certificate.
- Myth: Digital health records (EMR) make everything easier.
Reality: In many Indian hospitals, EMRs are used for billing, not for clinical notes. You must check if the clinical source data is actually in the EMR or still in handwritten folders.
Common Mistakes
Sponsor Mistakes
- Rushing the SQV to meet a quarterly “Site Selection” KPI.
- Ignoring “soft signals” like a PI who doesn’t show up on time for the qualification meeting.
- Underestimating the time required for India-specific approvals (ICMR/NOC for biological samples).
CRO Mistakes
- Sending junior CRAs with no therapeutic expertise to conduct the visit.
- Sugarcoating site weaknesses to ensure the sponsor selects the site.
- Failing to verify the workload of the study coordinator.
Site Mistakes
- Over-promising on recruitment to secure the study.
- Failing to disclose other competing trials in the same therapeutic area.
- Using a “standard” CV that hasn’t been updated in three years.
Counterintuitive Insight: Why “Busy” PIs Can Be a Red Flag
Most sponsors want the top-ranked doctor in the country. In India, these doctors often see 100+ patients a day. They have no time to read the protocol, let alone oversee the daily nuances of a complex Phase II study Clinical Trial SQV Process.
The best-performing sites are often led by “Mid-Career” PIs. These are investigators who are established enough to have patient volume but are still hungry enough to be personally involved in the research. They value the publication potential and the data quality. During the SQV, I look for the PI who asks me technical questions about the drug’s mechanism of action. That shows real interest Clinical Trial SQV Process.
Practical Sponsor Checklist
Feasibility Stage (Pre-SQV)
- Verify IEC/IRB registration on the CDSCO portal (SUGAM).
- Check CTRI for the PI’s current trial load.
- Request a preliminary patient recruitment funnel based on ICD codes.
Startup Stage (During SQV)
- Inspect the pharmacy’s temperature logs for the last 12 months, not just the last week.
- Interview the study coordinator on their understanding of SAE timelines (24-hour reporting).
- Inspect the archive room. Is it damp? Is it prone to pests? (Common issues in certain Indian regions).
- Confirm the hospital’s willingness to accept the Sponsor’s indemnity and compensation clauses.
Execution Stage (Post-SQV Assessment)
- Review the SQV report specifically for “Investigator Oversight” risks.
- Ensure all action items from the SQV are closed before the SIV is scheduled.
- Cross-reference site staff names with debarment lists.
Regulatory and Compliance Context
Navigating the Indian regulatory environment requires adherence to multiple bodies:
- CDSCO (Central Drugs Standard Control Organization): The primary regulatory body. All SQVs must ensure the site is prepared for potential CDSCO inspections.
- DCGI (Drugs Controller General of India): The authority that grants trial permissions.
- ICMR (Indian Council of Medical Research): Their ethical guidelines (2017) are often more detailed than ICH-GCP regarding vulnerable populations.
- New Drugs and Clinical Trials Rules (2019): This is the Bible for trial execution in India. It defines the legal requirements for site selection and PI responsibility.
For technical inquiries or site-specific feasibility in India, you can reach out directly at govindpawar@oxygenclinicaltrials.com or connect via LinkedIn.
Suggested Visuals for Deployment
- Workflow Diagram: The path from Feasibility -> SQV -> EC Approval -> CTRI Registration -> SIV.
- Timeline Comparison: A bar chart showing the difference in startup timelines between “Site with registered EC” vs “Site needing new EC registration.”
- Approval Funnel: A visual showing how 100 potential sites are narrowed down to 10 qualified sites through the SQV process.
External References
FAQ Section
1. How long does a typical SQV take in India? A thorough SQV should take a full working day (6–8 hours). Rushed 2-hour visits often miss critical infrastructure gaps or staff competency issues. If the PI is only available for 10 minutes, that is a data point in itself—it indicates they may not have time for the actual trial.
2. Can we use a central Ethics Committee for all sites in India? Under the 2019 Rules, for multi-center trials, each site must have its own IEC. However, one IEC can act as the “Lead EC” for the technical review. Every site must still ensure their local IEC is registered with the CDSCO.
3. What is the biggest reason for site rejection during an SQV? In my experience, it is a lack of dedicated research staff. If the site expects the hospital’s regular nursing staff to conduct study procedures without a dedicated coordinator, the risk of protocol deviations is too high to proceed.
4. How does Oxygen Clinical Trial help in the SQV process? We don’t just conduct a visit; we perform an operational audit. We look for the “hidden” problems—like a PI’s upcoming retirement, a hospital’s changing management, or a pharmacy that lacks proper access control. We bridge the gap between “paper feasibility” and “execution reality.”
5. What is the impact of ALCOA+ on the SQV? Sponsors are now looking for the “plus”—Complete, Consistent, Enduring, and Available. During the SQV, we evaluate the site’s record-keeping history. If their past studies show inconsistent source data, we flag them as high-risk regardless of their patient volume.
Selecting the right site in India is a strategic decision that determines the fate of your global data package. Don’t let a poorly executed SQV be the reason your trial fails. Focus on the people and the processes, not just the bricks and mortar. Contact us today to ensure your Indian sites are audit-ready