By Govind Pawar, Senior Clinical Operations Leader, Oxygen Clinical Research and Services
1. Introduction
Clinicaltrial Feasibility Workflow India. Over the past decade, India has moved from a peripheral recruitment hub to a core contributor in global Phase II‑IV trials. The shift is not because of incentives alone; it is the result of a mature regulatory framework, a growing pool of qualified investigators, and an expanding network of capable sites. However, the speed and predictability of a study still hinge on a single, often under‑estimated activity: site feasibility assessment.
In my 15 years of executing trials for Indian and global sponsors, I have seen sponsors either rush through feasibility and later confront protocol deviations, or spend months on exhaustive questionnaires only to lose the best sites to competing studies. The balance lies in a structured, data‑driven workflow that respects the Indian operational context while delivering the speed, compliance, and data quality sponsors demand.
The following guide walks a sponsor through the end‑to‑end feasibility workflow as practiced by Oxygen Clinical Research and Services and leading CROs in India. It is grounded in field observations, includes practical tables and checklists, and highlights the trade‑offs that rarely appear in vendor brochures.
2. Why Feasibility Is the First Gatekeeper
| Aspect | Typical Sponsor Expectation | Indian Reality (Observed) | Impact of Mismatch |
| Patient pool size | Census‑based estimates from epidemiology reports | Regional disease clustering, referral patterns, and socio‑economic barriers affect enrolment | Over‑recruitment targets, delayed timelines |
| Investigator experience | Number of published papers or past trial count | Hands‑on GCP training, site staff turnover, and local ethics committee (EC) timelines matter more | Protocol non‑compliance, protocol amendment requests |
| Infrastructure | Presence of a CTMS or electronic data capture (EDC) system | Variable EMR integration, intermittent power, and internet bandwidth | Data entry lag, increased monitoring effort |
| Regulatory turnaround | 30‑day EC approval quoted by many sites | EC meeting frequency (monthly vs quarterly), state‑level approvals, and CDSCO review for certain INDs | Unforeseen delays of 2‑4 weeks or more |
| Budget compliance | Fixed per‑patient cost | Local taxes, GST, site‑specific overheads, and compensation norms for patients and staff | Budget overruns, contract renegotiations |
The table underscores that feasibility is not a checklist exercise; each element interacts with the others. A robust workflow captures these interactions early, allowing sponsors to make informed trade‑offs between speed, cost, and data integrity.
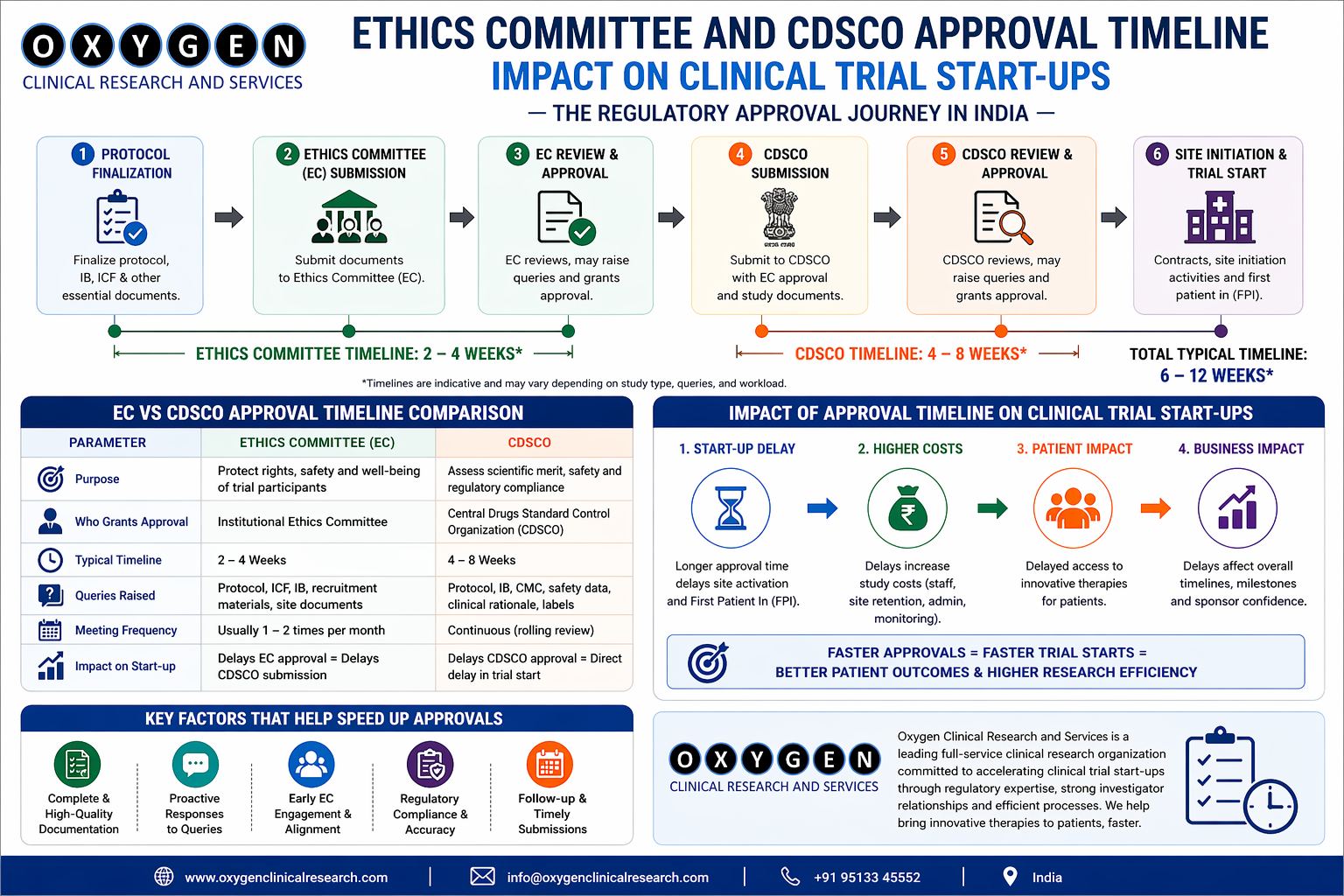
3. Indian Regulatory and Operational Context
- Regulatory Authorities – Central Drugs Standard Control Organization (CDSCO) governs IND/IDE approvals. For multi‑regional trials, the New Drug Advisory Committee (NDAC) review adds 2–3 weeks.
- Ethics Committees – Over 1,200 ECs operate under the ICMR guidelines. Their meeting frequency, quorum requirements, and conflict‑of‑interest policies vary widely.
- Governing Laws – The Drugs and Cosmetics Act (Amended 2019) mandates compensation for trial‑related injury. Sponsors must factor compensation reserves into the budget.
- Data Protection – Draft Personal Data Protection Bill (2024) introduces consent‑level data handling requirements. Sites without compliant processes may need additional SOPs.
- Taxation – GST at 18 % on services and 5 % on medical devices affect the overall site cost structure.
Understanding these layers is essential when designing the feasibility questionnaire and when interpreting site responses.
4. Step‑by‑Step Feasibility Workflow
Below is the workflow that Oxygen Clinical Research and Services follows for every new protocol. The steps are sequential but iterative; a failure at any point triggers a “go‑back” to the previous stage with a documented rationale.
4.1. Protocol Intake & Feasibility Scope Definition
| Action | Owner | Output |
| Review protocol synopsis, inclusion/exclusion criteria, and primary endpoints | Sponsor Clinical Lead | Feasibility Scope Document (FSD) |
| Identify therapeutic area experts within India | CRO Medical Lead | List of target investigator specialties |
| Set target enrolment per site, timeline, and budget envelope | Sponsor Finance & Operations | Feasibility Parameters Sheet |
4.2. Site Database Enrichment
| Data Source | Frequency of Refresh | Typical Lag |
| Internal CRO site master | Quarterly | ≤ 30 days |
| ICMR clinical trial registry | Monthly | ≤ 15 days |
| State health department enrollment reports | Bi‑annual | ≤ 90 days |
| Private hospital patient‑level EMR analytics | Real‑time (if API available) | Near‑real time |
Enrich the master list with the latest patient census, investigator turnover, and recent EC meeting minutes.
4.3. Preliminary Screening (Desk Review)
- Eligibility Matrix – Map each inclusion/exclusion criterion to the site’s disease registry.
- Infrastructure Scorecard – Rate EMR, pharmacy, laboratory, and imaging capabilities on a 1‑5 scale.
- Regulatory Red Flags – Flag sites with pending IND amendments or those in states with additional health‑department clearances.
Sites scoring < 3 on any critical element are either excluded or sent for targeted clarification.
4.4. Structured Feasibility Questionnaire
| Section | Sample Question | Rationale |
| Patient Availability | “Average number of new diagnosed patients per month for disease X in the last 12 months?” | Quantifies realistic pool |
| Investigator Commitment | “Will the PI be available for ≥ 75 % of monitoring visits?” | Predicts monitoring effort |
| Regulatory Timeline | “When is the next scheduled EC meeting?” | Estimates approval date |
| Site Staff Turnover | “Number of CRA replacements in the past 12 months?” | Anticipates operational continuity |
| Compensation & Tax | “What is the GST rate applied to sponsor payments?” | Budget alignment |
Distribute the questionnaire electronically, allowing sites to upload supporting documents (e.g., patient logs, EC minutes).
4.5. Data Verification & Site Visit
- Desk Verification – Cross‑check submitted data against hospital MIS, state health portals, or published registries.
- Virtual Walk‑through – Use video conferencing to inspect pharmacy storage, lab equipment, and data capture tools.
- On‑site Feasibility Audit (if needed) – Allocate a CRO Feasibility Auditor for high‑risk or high‑potential sites.
During the audit, capture process friction points (e.g., inconsistent SOPs for sample handling) and log them in the Feasibility Audit Report.
4.6. Feasibility Scoring & Decision Matrix
| Score Range | Decision |
| 85‑100 | Proceed to Site Initiation (SI) |
| 70‑84 | Conditional – require corrective actions (e.g., additional training) |
| < 70 | Reject or place on “future watch” list |
The scoring algorithm weighs patient pool (40 %), investigator experience (20 %), infrastructure (20 %), regulatory timeline (10 %), and budget compliance (10 %). Adjust weights for therapeutic area specifics (e.g., oncology may give patient pool 50 %).
4.7. Feasibility Report & Sponsor Sign‑off
The final deliverable includes:
- Site‑by‑site scorecards
- Risk register (patient recruitment, regulatory, data quality)
- Mitigation plan with owners and timelines
- Budget reconciliation worksheet
Sponsor sign‑off triggers the Site Initiation Pack (SIP) preparation.
5. Practical Checklists
5.1. Feasibility Questionnaire Checklist
- Disease incidence data for the last 12 months
- List of sub‑investigators and their GCP certifications
- Current EC meeting schedule and latest approval minutes
- Pharmacy cold chain capability (if biologics)
- Backup power source (UPS / generator) and average uptime
- GST registration certificate and invoicing template
5.2. On‑Site Audit Checklist
- Verify patient logbooks against EMR extracts
- Observe informed consent process with a mock patient
- Confirm availability of calibrated lab equipment (e.g., hematology analyzer)
- Review data entry workflow: paper CRF vs EDC
- Assess staff turnover through HR records for the past 12 months
6. Challenges & Mitigation Strategies
| Challenge | Why It Occurs | Mitigation |
| Inconsistent EC timelines | ECs meet quarterly; some require quorum of external members | Build a 2‑week buffer in the feasibility timeline; maintain a pre‑approved EC template to accelerate review |
| Patient migration to private hospitals | Public hospitals lose high‑income patients to private centers | Include private‑hospital sites in the same region; negotiate data‑sharing agreements |
| High staff turnover | Competitive market for clinical research coordinators (CRCs) | Offer site‑level training contracts (e.g., 12‑month CRC retention agreement) and performance‑based incentives |
| GST impact on site payments | Mis‑understanding of tax invoicing leads to delayed payments | Provide a standard GST‑compliant invoice template; clarify that the sponsor’s budget includes GST |
| Data protection compliance | New Personal Data Protection Bill requires consent logs | Integrate consent‑tracking module in EDC; conduct a site‑level data‑privacy audit before SI |
7. Myths vs. Reality
| Myth | Reality |
| “A site that has enrolled > 200 patients in the last year will automatically meet our enrolment target.” | Prior enrolment does not guarantee eligibility for the new protocol’s specific inclusion criteria. |
| “If the PI has published extensively, the site will deliver high‑quality data.” | Publication record does not reflect day‑to‑day GCP compliance; on‑site SOP adherence is a better predictor. |
| “All Indian ECs process submissions within 30 days.” | Many ECs operate on a monthly schedule; some require supplementary documents, extending the timeline to 45‑60 days. |
| “GST is the only tax to consider for site fees.” | Some states levy professional tax on consulting fees; sponsors must account for both at the contract stage. |
| “Virtual feasibility audits are sufficient for all sites.” | Rural sites may have limited internet bandwidth, leading to incomplete visual inspection; a hybrid approach (virtual + brief on‑site) is often required. |
8. Common Mistakes by Stakeholders
8.1. Sponsors
- Over‑reliance on a single “high‑volume” site without assessing its current workload Clinicaltrial Feasibility Workflow India.
- Ignoring the need for site‑specific budgeting for compensation and GST Clinicaltrial Feasibility Workflow India.
- Seting a blanket enrolment target without region‑wise patient pool analysis Clinicaltrial Feasibility Workflow India.
8.2. CROs
- Sending a generic questionnaire that does not capture therapeutic‑area nuances.
- Failing to update the master site list after each audit, leading to outdated feasibility data.
8.3. Site Management Professionals (SMPs)
- Providing optimistic patient numbers to win the contract, then under‑delivering.
- Not documenting regulatory correspondence, which later creates audit findings.
8.4. Investigators (PIs)
- Agreeing to recruitment targets beyond their realistic capacity, causing protocol deviations Clinicaltrial Feasibility Workflow India.
- Not involving sub‑investigators early in the feasibility discussion, leading to staffing gaps Clinicaltrial Feasibility Workflow India.
9. FAQ
| Sr. No. | Question | Answer |
| 1 | How many sites should I include in a Phase II oncology trial in India? | Typically 8‑12 sites, each capable of enrolling 8‑12 patients per month, based on a pooled recruitment capacity of 100‑120 patients/month. |
| 2 | What is the average EC approval time for a multi‑center study? | 30‑45 days for most Tier‑1 ECs; Tier‑2 ECs can take up to 60 days. |
| 3 | Do I need a separate IND amendment for each site? | No. A single IND amendment covering all Indian sites is sufficient, but state‑level clearances may be required for certain investigational products. |
| 4 | How is GST applied to sponsor payments? | GST is levied on the service fee component; the sponsor must issue a GST‑compliant invoice. The site’s GST registration number must be captured in the contract. |
| 5 | What are the typical patient compensation ranges? | For non‑interventional studies INR 2,000‑5,000 per visit; for interventional studies INR 5,000‑15,000 depending on procedure intensity. |
| 6 | Can I use a virtual site initiation to save time? | Yes, for sites with stable internet and EDC access, but an on‑site verification of pharmacy and lab storage is still recommended. |
| 7 | How do I handle sites that have a high turnover of CRCs? | Include a “staff continuity clause” in the site contract and budget for a 3‑month training overlap period. |
| 8 | What documentation is required for compensation of trial‑related injury? | A signed compensation agreement, the latest CDSCO compensation guideline, and the site’s insurance policy details. |
| 9 | Is it necessary to audit every site before enrolment? | Not mandatory. Prioritize audit for high‑risk sites (e.g., low infrastructure score, past compliance findings). |
| 10 | How does the Personal Data Protection Bill affect consent forms? | Consent forms must include a clause on data storage, sharing, and the right to withdraw consent for data use. A site‑level data‑privacy SOP is advisable. |
10. Actionable Conclusion
A well‑executed feasibility assessment in India is the single most effective lever for improving trial speed, predictability, compliance, and data quality. Sponsors should:
- Standardize the workflow – adopt the step‑by‑step process outlined above and embed it in the sponsor’s SOP library.
- Invest in data enrichment – maintain a live site master that